Foreword: The following article was written BC. Before Coronavirus. The information that follows needs to be temporarily viewed in a different light…
In the last few decades, personal cleanliness has been promoted intensely. One of the most recent marketable personal hygiene items is hand sanitizer.
People purchase and use the scented stuff in all manner of situations, but especially after they shake hands with someone they suspect is sick!
A typical sanitizer composition contains alcohol, a thickening agent, and scent.
Another popular hygiene product is antibacterial soap. This is so despite the widespread successful use of regular soap. People want the latest and greatest. Newer must be better, right? Yet antibacterial soaps have not been received altogether with open arms.
Antibacterial Soaps – Controversy, Big Time
The concept of an antimicrobial or antibacterial soap seems commendable. Yet, reality does not always measure up to concept. How does antibacterial soap measure up when put to the test?
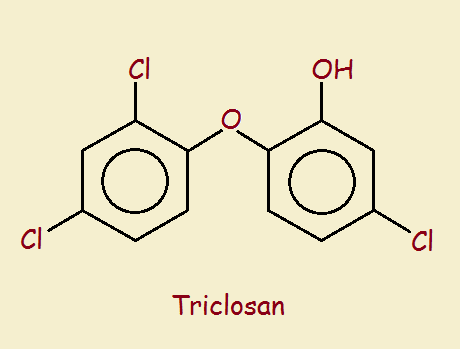
As the name suggests, antibacterial soaps include special ingredients designed to reduce the number of bacteria remaining on an object (often human skin) after its use. Businesses are inclined to favor established well-known and relatively inexpensive ingredients to accomplish any task. One such ingredient used in antibacterial soaps is triclosan.
There is much controversy over the use of triclosan. For one thing, although there may be some use for the ingredient in applications such as oral use in fighting plaque formation and gingivitis, much of what it is actually used for may be just as well-handled by ordinary soap and water or other already well-founded sanitation.
Triclosan is undergoing investigation concerning its suspected ill-effects to human health and environmental systems. For example, a marked percentage of triclosan passing into the sewer systems may actually be overpowering those systems and passing into bodies of water.
The author is a retired chemist who briefly worked at a sewer plant. He has noted over the years how highly chlorinated products have been used in fire retardation and pesticide use, and antibacterial products (for instance, hexachlorophene) and was not surprised to discover triclosan contains three chlorine atoms and two phenol rings. However, my perspective is nothing more than that. For an official perspective on triclosan, consider what the Food and Drug Administration writes concerning the general use of antibacterial soap (see 1st reference, below).
Note: You might also enjoy Shampoo – Is It Soap, Is It Detergent, or What?
References:
- FDA: Antibacterial Soap? You Can Skip It, Use Plain Soap and Water
- Smithsonian.com: Triclosan, a Chemical Used in Antibacterial Soaps, is Found to Impair Muscle Function
- Cosmetics and Skin: Hexachlorophene
← Back to Food and Health
← Home

I wash my hands after being near someone who has a cold or after touching handles or surfaces that they may have touched but I don’t worry about whether the hand cleanser is antibacterial. Plain soap and water is fine. The only exception for me is if I am accompanying my sister-in-law to a hospital visit, then I use the hand cleanser available, which probably contains alcohol and is a preventive against MRSA.