Benzene? What’s that?
Living creatures universally share an important characteristic: they all are constructed of carbon-containing compounds. For that reason, chemists call such compounds organic. Now the term organic has taken on additional meaning. Compounds that are similar, but not found in nature, are also called organic compounds.
One group of organic compounds, whether found in nature or not, possess special properties—chemical and physical—that put them into a category apart. Once it was thought such compounds were distinguishable by smell or aroma. Each was labeled an aromatic, and—the property itself—aromaticity.
Simple Hydrocarbons
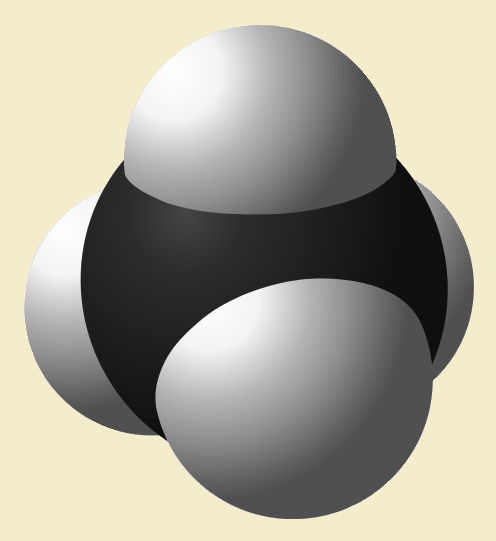
The simplest hydrocarbon1 is methane (CH4)—a gas. It possesses a tetrahedral structure, with a carbon atom at its center and four hydrogen atoms at the corners (See Figure 1). Since there are a total of four bonds—one to each of the hydrogen atoms—carbon here is said to have a valence of four. Most organic compounds feature valence-four carbon.
The next simplest hydrocarbon is ethane (C2H6). It is the equivalent of two methane molecules with one hydrogen atom removed from each, connected together, carbon bonded to carbon. The ability of carbon to bond to itself multiple times to create even lengthy chains, is a hallmark of carbon. Take normal hexane (C6H14)2. One way to write its structure, at length, is CH3CH2CH2CH2CH2CH3.
Hexane Becomes a Ring
Now imagine the leftmost carbon bonding to the rightmost carbon. In order to do that and maintain the carbon valence of four, one hydrogen atom on each end has to go. The result is the hexagonal structure, written simply C6H12, illustrated in Figure 2. This molecule, logically, is called cyclohexane. You will notice that the structure as drawn is not flat. This is because a tetrahedral angle is approximately 109.5°. Cyclohexane tends toward two forms: a chair form, and a boat form.
Now Remove Half the Hydrogen Atoms
If we remove half of those twelve hydrogen atoms, leaving only six—one attached to each carbon atom—we now have C6H6. This would appear to have upset the valence of our carbon atoms—but not so. Instead, we have introduced double bonds. This enables us to retain valence-four carbon.
Early on, this material was named 1,3,5-cyclohexatriene. How was this name chosen? If you draw a simple hexagon and label the apex carbon atom as 1, then go clockwise and write 2, 3, 4, 5, and 6, then label each double bond by the lower carbon number3 for each of the bonds, you get the 1,3,5-. The cyclo- refers to its being a ring structure. The tri- to the number of special bonds being three, and the -ene because those special bonds resemble the double bonds of an alkene.
Benzene – Special Features
Here’s a fascinating feature of this substance. If you write down 2,4,6-cyclohexatriene, at first it might look different from the 1,3,5-cyclohexatriene. But it’s not different—it’s the same thing. In fact, there are other structures that look a little strange and are beyond the scope of this article that can also be written down and represent the very same compound.4 These interchangeable structures are termed “resonance structures.” The sum total must represent the true structure of the aromatic hydrocarbon benzene.
Finally, the presence of the double bonds and their apparent equivalence means the structure should have 6 equivalent interior angles of 60°. Does it—yes, it does. And since they are all equal and the hexagon is regular, the molecule has the extra special feature of being flat. As we shall see in Part Two of this article, that makes all the difference in the world in defining the aromatic hydrocarbon benzene.
1 A hydrocarbon is any molecule consisting solely of carbon and hydrogen atoms.
2 Normal hexane or n-hexane, means the chain is straight, without side-branches.
3 If the three bonds are as follow: one between carbon atom 1 and 2, one between carbon atom 3 and 4, and one carbon atom between 5 and 6, using the lower numbers, you have the 1,3,5-structure.
4 The ability to draw multiple structures representing the same compound that helps chemists recognize the property of aromaticity.
[END of Part One] Proceed to Part Two
References:
- UCLA: All About the Chair Conformation [of cyclohexane]
← Back to Classic Science
← Home
