As the world became more sophisticated, a philosophy sprouted declaring the problems plaguing mankind would one day be resolved via education.
A popular slogan developed by one manufacturing giant suggested their products could assist one to better living through chemistry.
But what of organophosphates – potentially dangerous pesticides?
Better Living through Chemistry
In some ways, the world is a better place due to the aid of chemists. Perhaps, though, the opposite could be said with equal fervor. Chemistry has also degraded quality of life in some respects. You don’t have to think long to find an illustration of this – consider the use of phosgene and mustard gas during times of war.
In peaceful times as well, chemists unwittingly caused harm to the ecosystem. Think of the infamous pesticide DDT, sarcastically nicknamed Drop Dead Twice. With author Rachel Carson at the forefront, Society sought to outlaw the use of DDT.
But let us now turn our attention to organophosphates – simple organic esters of phosphoric acid, H₃PO₄.
Phosphoric Acid
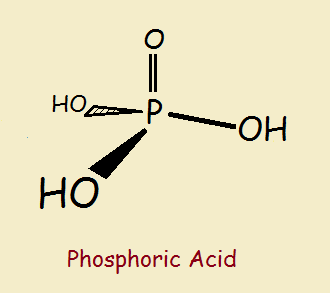
The tetrahedrally-shaped phosphoric acid is by itself an inorganic chemical, a so-called mineral acid. Although to the inorganic chemist, phosphoric acid is most often simply written H₃PO₄, this fails to reveal its chemical structure, illustrated at right.
Phosphoric acid has three hydrogen atoms that can ionize (gain electrical charges) or be replaced inorganically (such as by sodium atoms), or be replaced organically as in the formation of an ester. An ester is a compound formed by combining an alcohol molecule with a molecule of acid, with the loss of a water molecule.
We will consider phosphoric acid esters — organophosphates.
Carboxylic Acid Esters
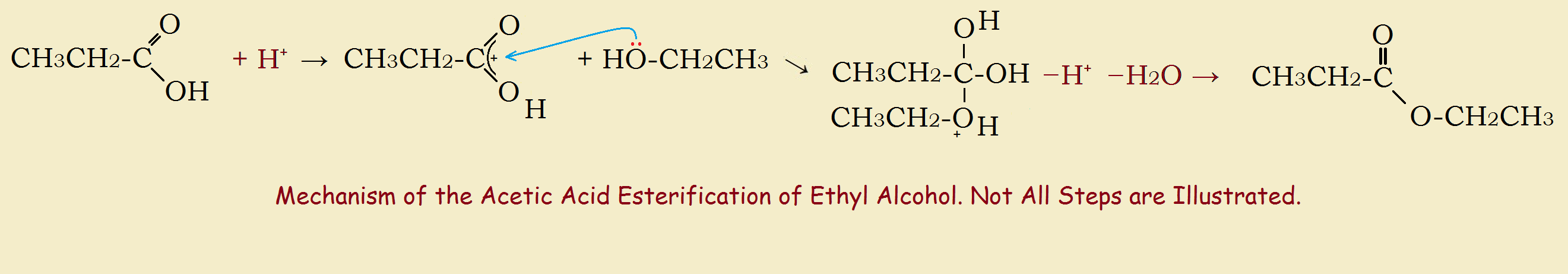
We will first introduce the formation of esters using carboxylic acids. One well-known ester used for many decades is ethyl acetate, so-called oily fingernail polish remover. Its formation can be written: acetic acid plus ethyl alcohol yields ethyl acetate plus water.
CH3CH2-COOH + CH3CH2-OH → CH3CH₂-CO-O-CH₂CH₃ + H₂O
The above reaction occurs in the presence of a stronger mineral acid. Sulfuric acid also dehydrates, removing one of the products, thus assuring completion of reaction. So it is frequently the acid of choice. For the mechanism, see the relevant image. The generic formula can be written,
R¹COOH + R²OH → R¹-COOR² + H2O
R¹ and R² can be different, or they can be (as in the example above) the same.
Phosphoric Acid Esters (Organophosphates)
Even in organophosphates, phosphoric acid loses its hydrogen atoms. Copyright Image by Vincent Summers.
The esterification of phosphoric acid can be accomplished using three different alcohols, R¹OH, R²OH, and R³OH.
We write,
O=P(OH)₃ + R¹OH → O=P(OR¹)(OH)₂
and
O=P(OR¹)(OH)₂ + R²OH → O=P(OR¹)(OR²)(OH)
and
O=P(OR¹)(OR²)(OH) + R³OH → O=P(OR¹)(OR²)(OR³)
An example of a simple organophosphate of this sort, we will choose methyl alcohol for R¹OH, ethyl alcohol for R²OH, and n-propyl alcohol for R³OH. The structure of this even this very simple organophosphate is written,
O=P(OCH₃)(OCH₂CH₃)(OCH₂CH₂CH₃)
As might be imagined, nomenclature of the organophosphates can be very complex. Is it any surprise many prefer common names? Sometimes an organophosphate can be named as if it was a simple phosphate. One example is triethylphosphate, which is not used as a pesticide, but can serve as an intermediate in the manufacture of pesticides, and serves a host of other uses.
O=P(CH₃CH₂)₃
As in most areas of life, there can be both good and bad uses of just about anything. So, too, for organophosphates. Many organophosphates serve or have served as effective pesticides. An older CDC historical document reads, in part,
“What happens to organophosphates when they enter the environment?
“Because organophosphates are often sprayed on crops and plants, small particles of the chemical may be carried away from the field or yard before falling to the ground.
“After organophosphates are applied, they may be present in the soil, surface waters, and on the surface of the plants. They can move through the soil and contaminate ground water.
“Rain can wash organophosphates on soil and plant surfaces into surface waters.
“Organophosphates are rapidly broken down into other chemicals so they do not build up in the environment.
“Organophosphates are not likely to build up to high or dangerous levels in animal or plant foods that you might eat.”
Perhaps the main risk of organophosphates exposure is for workers in their manufacture. There has been some concern these substances may be linked to lymphoma and leukemia, as sometimes sufferers have had organophosphate metabolites (decomposition products) in their urine, most notably of chlorpyrifos and diazinon. Diazinon is one pesticide the author of this article has personally used on occasion.
Restricted Organophosphates
Some organophosphates are in current use, accomplishing the job for which they were developed. Time will reveal any causes for concern. Some organophosphates are restricted in their use, such as the chlorpyrifos previously cited. Others are no longer considered acceptable for use in many countries, such as parathion. Parathion, it should be noted, has the oxygen doubly bonded to phosphorus, replaced by a sulfur atom. Hence the suffix –thion.
So: Better Living through Chemistry?
Our theme asks the question, are organophosphates another positive indicator that one can achieve better living through chemistry? As is almost always the case, the answer is a qualified one. Yes, it can be considered further evidence one can live better through chemistry. But only sometimes, and then, only to a degree.
Sometimes chemistry actually worsens life. Have you ever heard of the relatively modern organophosphate derivative – the warfare nerve gas, Sarin? How many of those who were exposed to the dreadful sarin would have been willing to utter the words “Better Living through Chemistry?”
Note: You might also enjoy The Truck Jake Brake – Quiet Helpful Technology?
References:
- Chemguide. The Mechanism for the Esterification Reaction
- Centers for Disease Control (CDC): . Environmental Hazards & Health Effects: Cancer Clusters
- Environmental Protection Agency (EPA). Organophosphates
