Human and animal life requires an exchange of oxygen (O2) and carbon dioxide (CO2). If the atmosphere contains more than a minimum of carbon monoxide (CO), life is at risk. Why? Consider the properties that distinguish carbon dioxide from the monoxide.
Carbon Dioxide
Carbon dioxide contains two atoms of oxygen bonded to one atom of carbon. The structure is written O=C=O. The length of its double bonds is 116.3 picometers. Bonding is covalent, not ionic. Water solubility at 250C is 1.45 g/l.
Carbon Monoxide
Carbon monoxide is written simply C≡O. The triple bond is 112.8 picometers. It consists of two polar covalent bonds plus a coordinate covalent bond. Its dissociation energy is 1072 kilojoules per mole. Water solubility at 250 Celsius is 27.6 mg/l.
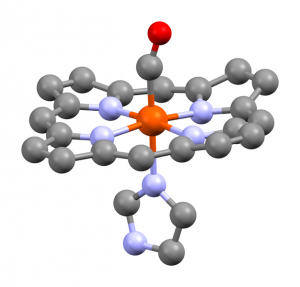
The affinity of carbon monoxide for transition metals (such as nickel and iron) is of special interest. For instance, they can form carbonyls. Among the best known are nickel tetracarbonyl, Ni(CO)4, and iron pentacarbonyl, Fe(CO)5.
Metal carbonyls bond a number of ways, including sigma and pi back bonding. Carbonyls feature stabilizing bond resonances. All of these involve unfilled d-orbitals of the transition metals.
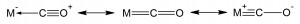
Carbon Dioxide and Hemoglobin
Although bonding between carbon dioxide and hemoglobin occurs, it is easily reversed. It is part of respiration. It merely lowers the affinity of hemoglobin for oxygen. Carbon monoxide bonds with hemoglobin to a potentially fatal degree.
Combining CO with Hemoglobin
Monoxide bonds to the iron found in hemoglobin. It is the oxygen carrying component of blood. Bonds to carbon monoxide are 200+ times stronger than bonds to oxygen. Once hemoglobin bonds to CO, it is hours before it is available to bond with oxygen. What if a sizable percentage of hemoglobin bonds to monoxide? Death results by asphyxiation.
Note: You might also enjoy Hydrogen Sulfide or Hydrogen Cyanide: Which is More Dangerous?
References:

Yes, a lot of detective and medical mystery books have been written using carbon monoxide as the agent of death.