 What does corncob destructive distillation produce? A most interesting product—furfural. What is it? What is it good for?
What does corncob destructive distillation produce? A most interesting product—furfural. What is it? What is it good for?
What is Furfural?
Furfural is an interesting organic compound, possessing a five member ring structure with a heterocyclic atom (oxygen) and several functional groups built in. It is an ether, a diene, and an aldehyde.
What is the Reaction?
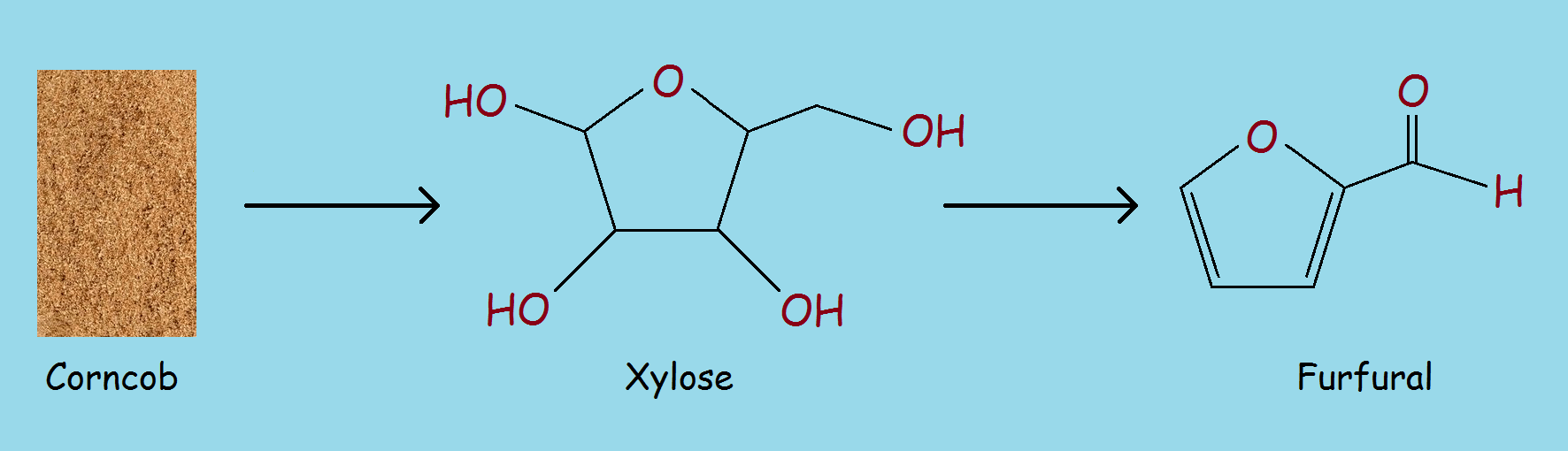
Corncobs1 are rich in hemicellulose. Hemicellulose can be broken down to produce the five-carbon sugar—xylose, C5H10O5. Xylose can react further to produce the heterocyclic aldehyde furfural, IUPAC2 name furan-2-carbaldehyde.
Corncobs (hemicellulose) → C5H10O5 → furfural (see diagram)
The steps can be combined. The resultant reaction process is termed destructive distillation.
Corncob Destructive Distillation
The corncobs may be dried and chopped into tiny bits the size of a grain of corn or smaller. These may be combined with sodium chloride and sulfuric acid, refluxed using a condenser, and afterward, distilled. Additional processing follows, but this is the basic methodology. The acid breaks down (hydrolyzes) larger molecules, dehydrating them and modifying their structure.
Why Bother?
So what is the value of furfural that it should be manufactured in this fashion? It is used in agricultural applications and as a non-toxic solvent. Furfural can be converted into a variety of useful related compounds, including tetrahydrofuran (THF) derivatives. More succinctly, as Biomass Magazine informs us, furfural is an appealing “alternative for the production of everything from antacids and fertilizers to plastics and paints.”
1 Other cellulose-rich byproducts can be used, such as sugarcane bagasse, oat hulls, peanut husks and sawdust.
2 International Union of Pure and Applied Chemistry
Note: You might also enjoy Terpenes and Terpenoids: Isoprene Rule
References:
- ACS: The Preparation of Xylose from Corn Cobs
- Organic Syntheses: Furfural (2-furaldehyde)
- Biomass Magazine: Furfural: Future Feedstock for Fuels and Chemicals
