
Global warming is a multi-faceted problem. It even involves chemistry.
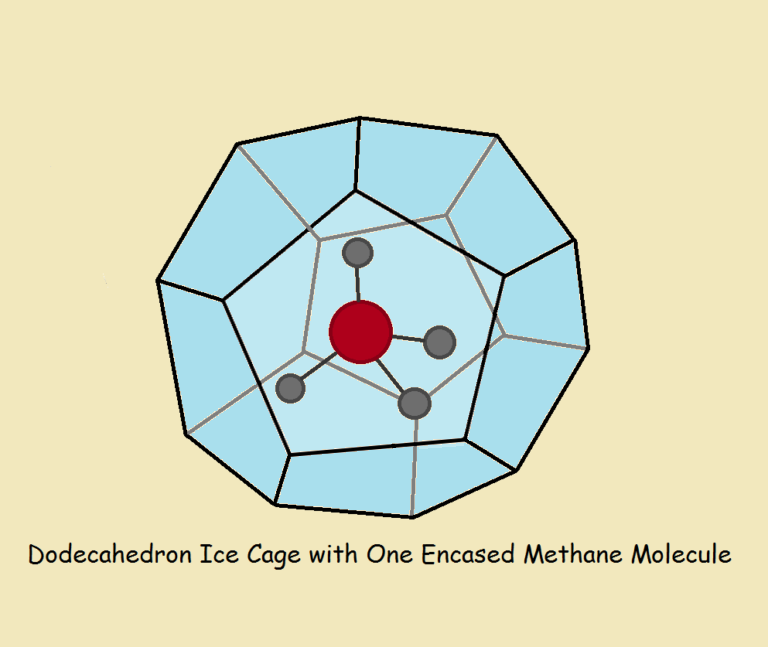
A clathrate is an inclusion complex. This is a kind of compound in which a molecule is completely enclosed by another substance.
It is for this reason that a clathrate is at times called a “cage compound.”
Global Warming
Perhaps the most important clathrate is a molecule of methane gas (CH₄) entrapped in crystallized water – ice. This clathrate is found deep within the sea. It may play a role in terrifying events connected to global warming.
Many (perhaps most) opponents of global warming are not scientists at all. So they may not know how to recognize how the most subtle changes can influence and trigger major events. This should not surprise us. The world is controlled, after all, by the almighty buck. And the almighty buck leads us back to politics. People often believe what they want to believe.
 A Tenuous Existence
A Tenuous Existence
Clathrate compounds require very specific circumstances in order to form. The conditions required are readily found deep within the sea. Even a fractional degree of temperature rise could initiate the melting of barely frozen methane water clathrates, potentiating seismic events.
This could lead to more severe storms that include larger and more horrific hurricanes. In fact, even earthquakes and tsunamis.
Note: You might also enjoy Maximize Carbon Dioxide Greenhouse Gas Production
References:

Are clathrates free in the water, even if at the bottom of a deep trench or are they contained in rock? There have been stories of boats overturned by giant bubbles of methane, could these stories be related to clathrates? Fire ice? Can a clathrate exist at sea level temperatures and pressures, even if only long enough to burn?
It does not appear they are in rock, but develop in locations offering the appropriate conditions. In other words, I don’t know. Here are a couple of brief statements taken from Wikipedia… 1. Clathrates have been found to occur naturally in large quantities. Around 6.4 trillion (6.4×10^12) tonnes of methane is trapped in deposits of methane clathrate on the deep ocean floor. Such deposits can be found on the Norwegian continental shelf in the northern headwall flank of the Storegga Slide. Clathrates can also exist as permafrost, as at the Mallik gas hydrate site in the Mackenzie Delta of northwestern Canadian Arctic. and 2. Naturally on Earth gas hydrates can be found on the seabed, in ocean sediments, in deep lake sediments (e.g. Lake Baikal), as well as in the permafrost regions. The amount of methane potentially trapped in natural methane hydrate deposits may be significant (1015 to 1017 cubic metres), which makes them of major interest as a potential energy resource. Catastrophic release of methane from the decomposition of such deposits may lead to a global climate change, because CH4 is more of an efficient greenhouse gas than CO2 (see Atmospheric methane).
It would then be better to burn the methane off, to produce carbon dioxide and water?
It would be best not to warm the earth and make it a problem. There really is no good alternative.