
Gutta percha thermoplastic1 is a tough and leathery resinous produced from the milky fluid tapped from certain trees.
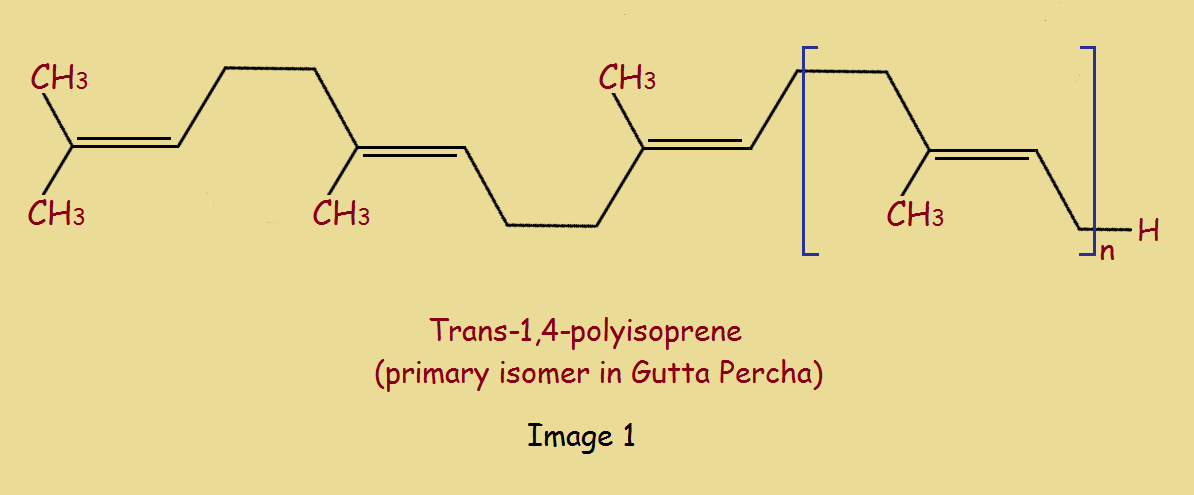
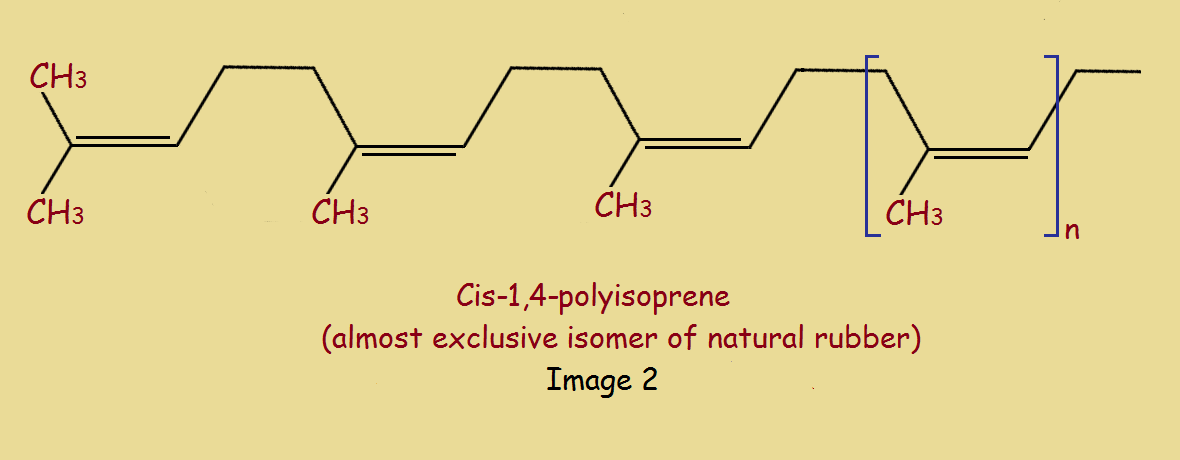
Isoprene is an extremely important building block widespread in nature. The main component of gutta percha is, in fact, the trans-1,4-isomer of polyisoprene. The cis-1,4,-isomer is, interestingly enough, the primary constituent of natural rubber. It is produced from the milky fluid tapped from “other” trees.
The reason for the considerable difference in physical characteristics between rubber and gutta percha (or, rather the trans and cis isomers of polypropylene) is the greater crystalline character of the trans isomer.
Notice the difference between the trans and cis isomers in Image 2. How do these isomers differ?
Synthesis from Isoprene
Trans-1,4-polyisoprene can be produced synthetically using Ziegler-Natta catalysts, yielding a completely man-made product. What is meant by a Ziegler-Natta catalyst? They are metal-based catalysts used to polymerize terminal alkenes—alkenes with a vinyl double bond—as for example, isoprene.
Let’s choose a general expression for this…
n CH2=CHR → –[CHR–CH2] n–
In our case, the compound at left would be isoprene and the result at right is Trans-1,4-polyisoprene.
Underwater Cables
Since gutta percha is thermoplastic and it is an elastomer2 and it is impervious to water, it was a natural choice to protect the Trans-Atlantic underwater cable. Events were favorable for its use, as well. The newly formed Gutta Percha Company was established in 1845. Perfect timing.
Golf Balls
It appears golf balls progressed from hair-stuffed and feather-stuffed leather to gutta percha. However, the so-called gutties lost favor by 1900. They were made, after all, of solid polymer! The outer covering or skin for future ball designs continued to be the tough and strong thermoplastic.
Use in Dentistry
The dentist and gutta percha came together in 1847, when it was introduced as a filling material. However, it was in 1867 it began to be used for root canal work. By and large, it was an excellent material. Nevertheless, there have been failures when using the polymer. Consider the following video on methods considered likely to result in a successful outcome.
Updating Gutta Percha in Dentistry
There is ongoing research in the improvement of gutta percha for dental root canal work. Most interestingly, one of those improvements involves the combining of the polymer with amoxicillin and nano diamonds. See the relevant reference, below.
1 A thermoplastic softens when heated, then hardens once again upon cooling.
2 An elastomer is a polymer with elastic properties. In this our thermoplastic is not completely unlike rubber.
Note: You might also enjoy What is Carrageenan?
References:
- Encyclopaedia Britannica: Polyisoprene
- History of the Atlantic Cable & Undersea Communications
- Scottish Golf History: History of Early Golf Balls
- Endoexperience.com: A Brief History of Endodontics
- ACS Publications: Nanodiamond–Gutta Percha Composite Biomaterials for Root Canal Therapy
