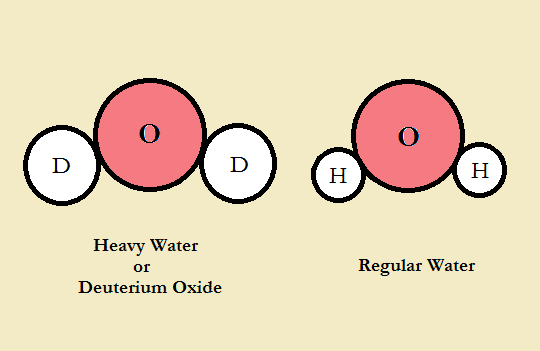
Hydrogen exists as three varieties or isotopes: protium, deuterium, and tritium. Heavy water, or deuterium oxide (D₂O) is the same compound as ordinary or tap water (H₂O) with one structure difference. Most of the hydrogen atoms in tap water have zero neutrons in their centers, or nuclei. On the other hand, every atom of deuterium contains precisely one neutron. We will not discuss tritium, with its one proton and two neutrons.
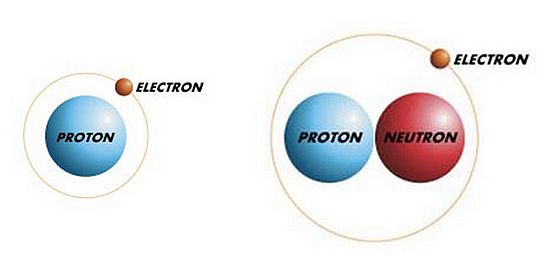
A Closer Inspection of Hydrogen Isotopes
The mass of an atom is determined by the nucleus. Since the weight of a proton and that of a neutron are nearly identical, deuterium atoms have a mass essentially double that of ordinary hydrogen. Hence one name for deuterium oxide is heavy water.
Heavy Water Laboratory Observations
Alan Soper of Rutherford Appleton Laboratory has found that the D-O bonds in heavy water are about 3% shorter than H-O bonds. A shorter bond is a stronger bond. This stronger direct D-O bonding in deuterium oxide logically decreases the strength of deuterium to oxygen intermolecular bonds (bonds between different molecules). They are about 4% longer than intermolecular H-O bonds.
Linearity – Going Straight
The D-O-D bond angle appears to be about 106° in the liquid phase. This makes heavy water a slightly more linear structure than ordinary water at 104.5°. The number of intermolecular hydrogen bonds for D₂O are somewhat fewer in number than for H₂O. Heavy water is denser than regular water. Finally, the freezing point of deuterium oxide is 4°C and it boils at 101°C.
Final Comments
Interestingly, intermolecular hydrogen bonding is a major factor in H₂O being a liquid rather than a gas at ordinary temperatures. Heavy water has fewer and weaker hydrogen bonds, so one might expect it to have a lower boiling point. However, the increased mass, coupled with influences from its other physical differences actually raises its boiling point a little higher than regular water.
A practical joke some find amusing is to put deuterium ice cubes in a glass of water. After asking those who are present if ice sinks or floats, and receiving the answer “it floats,” he or she then puts a heavy water ice cube in a glass and the cube plummets to the bottom of the glass.
Note: You might also enjoy “Three Hydrogen Isotopes: Protium, Deuterium, Tritium”
References:
- Argonne National Laboratory: Quantum Physics Makes Water Different (August 2008)
- D-O-D Bond Angle: K. Ichikawa, Y. Kameda, T. Yamaguchi, H. Wakita and M. Misawa, Neutron-diffraction investigation of the intramolecular structure of a water molecule in the liquid-phase at high-temperatures, Mol. Phys. 73 (1991) 79-86.
← Back to Quirky Science Nuggets
← Home

Heavy water is odd stuff. I heard of it many years ago in school but I seem to remember being told that if you watered plants solely with heavy water they would die. I have no idea whether that is true or not. Any idea?
I have read something to that effect. However (and oddly enough) it is considered quite likely humans could drink a fair amount of the stuff and not seriously suffer, though only a fair amount. Too much and humans, too, would suffer greatly.