
Orange jewelweed (Impatiens capensis)1 commonly grows in ditches along roadsides at the edges of moist forests. Lore has it that the sap of this plant rubbed onto poison ivy rash quickly alleviates the itching.
Could there be a scientific basis for this popular notion? Perhaps. Let’s begin our consideration by discussing the active principle in both poison ivy and jewelweed.
Poison Ivy and Urushiol
Urushiol is not a single compound, but a mixture of similar compounds that have the generic chemical structure show in the image associated with this article. R represents an alkyl chain that typically contains 15 to 17 carbon atoms that contain varying numbers of double bonds. The greater the number of double bonds, typically the more virulent the urushiol fraction.
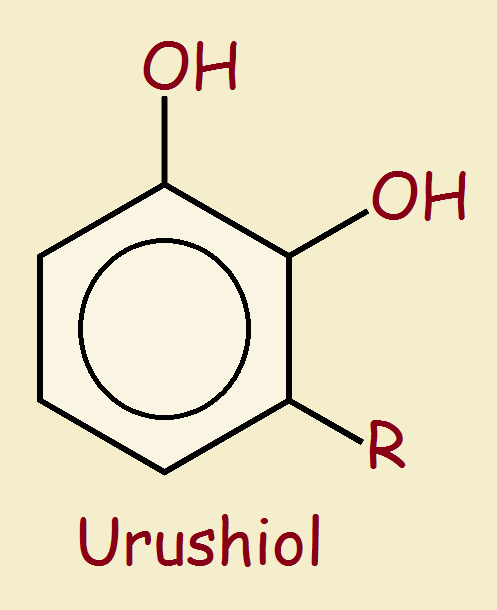 Urushiol is a polar covalent compound. This means it and all its component parts are insoluble in water, yet can form hydrogen bonds.
Urushiol is a polar covalent compound. This means it and all its component parts are insoluble in water, yet can form hydrogen bonds.
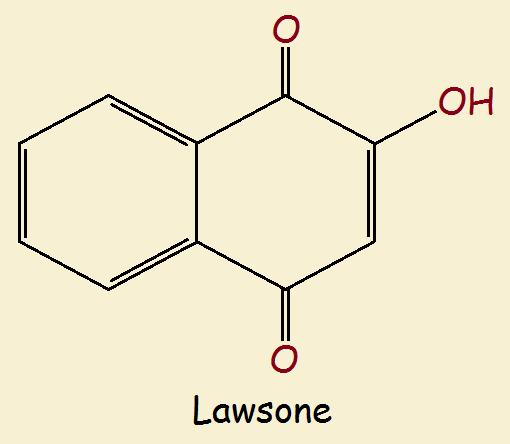
Jewelweed and Lawsone
Lawsone is the principal and possibly active ingredient in jewelweed, and is a dye. Due to its prevalence in henna, it is also called hennatannic acid. Lawsone has been used in temporary tattoos and in dying hair, although its use is controlled, even forbidden in some nations. Hennatannic acid is a naphthoquinone derivative and, like urushiol, is a polar covalent species.
 Could It Be?
Could It Be?
A general truism in chemistry is that like dissolves like. Urushiol and lawsone are both covalent, not ionic. They are both capable of forming hydrogen bonds. Could it be that lawsone in jewelweed sap, when applied to a fresh exposure of urushiol, hydrogen bonds to the urushiol and forms a kind of oily mix with it that can be partly wiped away? While it may not completely eliminate a rash from poison ivy exposure, might it not at least attenuate it so the sufferer gains some sense of relief?
1 There is a yellow variety of jewelweed, Impatiens pallida.
Note: You might also enjoy Hydrogen Bonding Effects at Various Levels
References:
- The Henna Page: Lawsone, by Dr. Boyan Bonev
- Penn State University – New Kensington Trail Species Pages
- European Commission: Opinion of the Scientific Committee… Lawsone
- Chemical & Engineering News Abstracts: Detecting Fingerprints with Lawsone

Interesting explanation. That might be something that a pharma firm would investigate. After all, if they could produce an item that could alleviate even part of the suffering from poison ivy, no doubt they would be able to make some money on that. It’s the kind of item that families would keep in a first aid box “just in case”.
Another thought concerning why jewelweed could cancel the effects of poison ivy are expressed in a book that says, “Dr. Rosen [a chemist at Rutgers University, New Brunswick says lawsone] binds to the same molecular site on the skin as urushiol. If applied quickly after contact with a poison plant, lawsone beats the urushiol to those sites, in effect locking it out. The simple result is that you don’t get the rash.”
I was just exploring this about jewelweed this week (have known of it for 40 years) while searching for information on the quinone component for use as a naturally occurring fungicide; which has been synthesized as a quinone based fungicide commercially. Mean while there were quite a lot of replicated studies referenced with jewelweed and poison ivy. Some showed little effect, others demonstrated a definite effect, but none showed any method that you could rely on being highly efficacious. Just google it if you want to explore it more.
I had heard what were, to me at the time, unproven rumors… wives’ tales. But the time arrived when the pro argument seemed to tip the scales the other way.
I was in my early 70s when I had my very first reaction to poison ivy. And it was a horrible experience! The Boy Scout Handbook taught me that one could be immune to poison ivy, but that one’s immunity could change overnight. Although I was a Boy Scout and had their “bible”, The Boy Scout Handbook, and I was a life-long outdoorsman who read all kinds of outdoor magazines, I had never, ever heard of “poison ivy soap” or “jewelweed salve” until my misery shoved me full-force into the bowels of the internet to find out about how to get relief from an outbreak of poison ivy. It was then that I learned about jewelweed and its active ingredient, lawsone, which negated the misery-causing urushiol found in poison ivy. Why hadn’t I heard of this before!? Perhaps because jewelweed and its active, urushiol-negating component, lawsone, are natural ingredients and as such, Big Pharma can’t patent it and make billions!! I discovered this fact after almost two weeks of sleepless nights due to my entire torso and thighs itching like crazy. Once educated, I went to my closest ACE Hardware store and bought a small bar of Grandma’s Poison Ivy Soap. I went home and took a shower using the soap and that was the first time in many days that I got any relief from the pain and itching of my very first reaction to poison ivy contact. At night, to better ensure a good night’s sleep, I’d again shower using the poison ivy soap. Ahhh, sweet relief and even sweeter night-long sleep. I now have a bar of poison ivy soap in each vehicle, my camping trailer, and in my medicine cabinet. Every home in America should have at least one bar of poison ivy soap at their command to quickly and effectively halt the further misery caused by poison ivy contact. If one knows they have touched, or have been in areas with poison ivy, taking a bath or shower using poison ivy soap shoud be their first order of business in order to mitigate further spread of the rash. You’re Welcome!