 To detect aroma, airborne chemicals must enter into our nasal passages. Volatile aromatic oils carried in when we breathe, interact with receptors located along those passages.
To detect aroma, airborne chemicals must enter into our nasal passages. Volatile aromatic oils carried in when we breathe, interact with receptors located along those passages.
Though flavor is somewhat influenced by our sense of smell, flavor primarily requires substance solubility in order to reach the different kinds of receptors located in various parts of the tongue. Let’s explore a little lemon chemistry.
Lemon Chemistry: Flavor
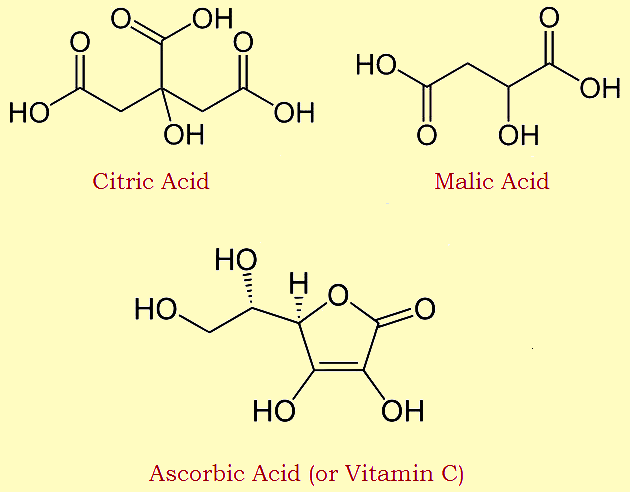
The tongue detects four, perhaps five, flavors. They are sweet, salty, sour, and bitter. Some add savory1. There can be no doubt the primary taste of lemon is sour. Mention sour and the organic chemist thinks of organic acids. Lemons are rich in three organic acids: citric, malic, and ascorbic (AKA Vitamin C). Citric acid predominates. Suck on citric acid and you’ll think of lemons.
Lemon Chemistry: Aroma
The odor of a lemon is a more complex matter. Much of its smell is located in the peel or zest. For this reason, zest is commonly used in gourmet cooking. But lemon peels are also of historical importance to furniture aficionados.
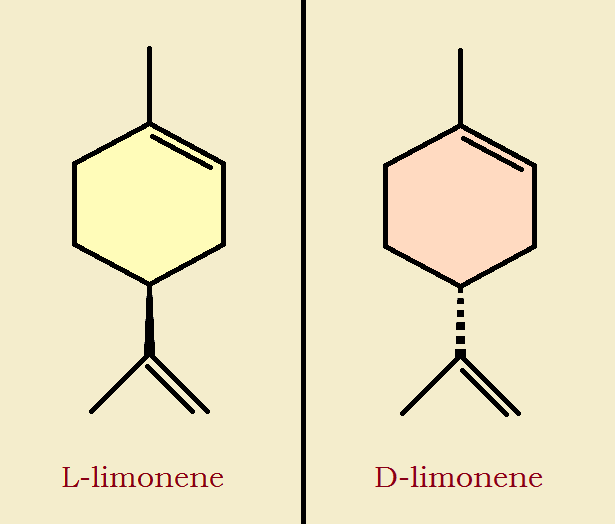
Many are familiar with lemon oil in furniture treatment compositions. Aromatic oil from the lemon includes and terpenes such as L-limonene2. D-limonene, the stereoisomer of L-limonene distinguishes the aroma of oranges. Additional fragrant compounds include certain coumarins.
Unfortunately, today much “lemon oil” is not really lemon oil. It often is, as the Smithsonian Institution suggests, “mineral oil with colorants and perfumes added.”
Did You Know?
What exactly are terpenes? They are a class of organic compounds or structures that can be built from the building block, isoprene2. The reader may note the similarity between the words isoprene and neoprene. Chlorinated isoprene or chloroprene was used to produce artificial rubber or neoprene during WW2. In fact, some researchers, even today, seek a commercially feasible process for reclaiming limonene by vacuum pyrolysis of used tires.
Concluding Remarks

As is suggested above, the lemon aroma ingredient limonene is of great importance. It not only imparts the fragrance of the lemon in cooking and deodorants, it is useful in the treatment of furniture. It is also a highly valued “natural” cleansing agent to replace more toxic cleansers in general purpose use. Practical results from showing an interest in lemon chemistry suggests we avidly pursue the chemistry of every living thing in the ecosystem.
1 Or, umami.
2 For further information concerning terpenes and the isoprene rule, see Terpenes and Terpenoids: Isoprene Rule
Note: You might also enjoy War, France, and Chicory – The Little Blue Flower Along the Road
References:
- The Chemistry of a Lemon
- ACS: Volatile Components of Peel and Leaf Oils…
- Smithsonian Institute: Smell the Difference
- Harvard University: The Bittersweet Truth of Sweet and Bitter Taste Receptors
- Journal of the American Chemical Society: Chemical Composition of Lemon Oil…
- Materials Recycling World: Journal of Analytical and Applied Pyrolysis

My husband uses lemon oil for cleaning his guitar.
I have a lemon tree that bears no fruit but the leaves seem to be full of the stuff that makes a lemon taste and smell – well – like a lemon.
Lemon is great in cooking. I love ginger and lemon tea, especially in the winter or when I have a cold or sore throat. I squeeze the lemon and keep the juice to one side, then pour boiling water over the lemon peel and some slices of fresh ginger and leave to steep for a while. Then I add in the lemon juice and drink. One lemon and a few slices of ginger makes enough for 1 litre of the good stuff. Occasionally I add honey but it’s not necessary.
I’m pretty sure that because of the rotation possible on carbon no. 4 on the cyclohexene the two pictures you have used for the two enantiomers are the same molecule, the mirror image should have the propyl group on 4 recessed into the page using the dotted line convention.
All the best
Point well-taken. Some draw that group with the dashed line notation, others draw the hydrogen (which I have not shown) in that fashion. Either would indicate the difference. I will adjust the image, accordingly.
Can lemon makes Coke explode?
Perhaps you are thinking of the “exploding” combination of Diet Coke and Mentos?