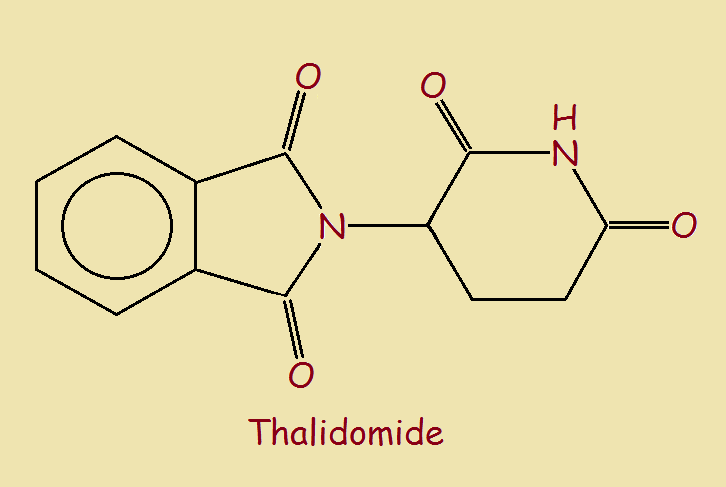
 Available to the general public in 1957, thalidomide1 was initially prescribed as a sedative or hypnotic medication. A sedative, we know, is intended to “calm the nerves”. What is an hypnotic? A number of online definitions suggest an hypnotic is a drug, likened to hypnotism, tending to produce sleep.
Available to the general public in 1957, thalidomide1 was initially prescribed as a sedative or hypnotic medication. A sedative, we know, is intended to “calm the nerves”. What is an hypnotic? A number of online definitions suggest an hypnotic is a drug, likened to hypnotism, tending to produce sleep.
A little later, it was given to fight nausea, and was prescribed to expectant mothers who experienced morning sickness. Who knew the morning sickness medication thalidomide would cause devastation to the babies? Search the web by entering the word thalidomide, and choosing the category images, and you will see quite a few photos of former “thalidomiders”.
A full-term pregnancy lasts nine months. While expectant mothers may have been calmed and their nausea lessened, it didn’t take long for thalidomide to produce devastating results… thousands of severely deformed babies, limbless or stumped limbs, roughly half of whom died. How shocking! What a tragedy! Short of death and loss of limbs, other severe medical issues were experienced by some, including blindness, deafness, heart damage, and malformation of passageways in the body.
Not counting war, it has been called “the greatest manmade global disaster” [Thalidomide Society].
Does the above video not suggest one should not automatically accept everything scientists and the pharmaceutical world offer without question? And take note that, after such a large blunder, providers fought compensation to the innocent victims.
In all fairness, the issue of thalidomide was not so much an issue of the doctors or their surgeries. It was primarily an issue of governments and the pharmaceutical industry. If governments had required ample testing, would there have been a problem? Even if there would have been, when once a problem was brought to the attention of manufacturers, was it properly handled?
The Question Arises…
Is thalidomide still being manufactured? Is it still being prescribed today? Surprisingly, yes, it is. But it is very carefully dispensed for only certain specific medical situations. Not only that, but it is illegal to dispense it in the U.S. to one who is pregnant, nursing, and (if the individual can become pregnant) on contraceptives.
What is Thalidomide Used For?
For some cases of leprosy
Chemical & Engineering News, cited below, says “thalidomide has potent anti-inflammatory effects that can ease erythema nodosum leprosum, a painful inflammatory condition associated with… leprosy. In July 1998, FDA approved the application of Celgene to market [Thalomid]… for treatment of that condition.”
How was this use discovered? Thalidomide was used by a physician in Jerusalem during the time it was not legally authorized. The result was favorable. It was legalized.
To fight multiple myeloma
Multiple myeloma is a cancer of plasma cells. DataPharm eMC informs us, “Thalidomide Celgene in combination with melphalan and prednisone is indicated as first line treatment of patients with untreated multiple myeloma, aged ≥ 65 years or ineligible for high dose chemotherapy.”
Does This Seem “Immoral”?
If the continued use of this drug seems just wrong, downright immoral, recall that most drugs used in less than miniscule quantities, are poisons. Doses of most medications are small. Not given for the correct application, even the most beneficial medications can cause severe problems. As an example, what would happen if the wonder drug insulin had been given to a person suffering hypoglycemia? Thalidomide is not just another jar off a chemist’s shelf. It was developed to exhibit certain characteristics.
It was ill-advised to expose a developing fetus to the drug, that goes without question. There are still former thalidomide babies in various countries who remind us just how devastating such use was. But there are other individuals, sick individuals who are not babies, that have derived benefit from the deadly “morning sickness” pill we call thalidomide.
1 Or thalomid.
Note: You might also enjoy Dangerous Medicine Including Foot Fluoroscope (X-Ray) Machines
References:
- The Guardian: My thalidomide family: Every time I went home I was a stranger
- Food and Drug Administration: U.S. Thalomid Label (Revised 2017)
- Chemical & Engineering News: Thalidomide
- DataPharm eMC: Thalidomide Celgene 50 mg Hard Capsules

The thalidomide tragedy was a triumph of greed over commonsense. BigPharma only gets money when it gets a new drug to market, so it does everything in its power to shortcut the process of testing and analysing. Many new drugs are useful for certain situations but always the mantra “Safe rather than sorry” must be applied.
Morning sickness is not a disease. It doesn’t last [as] many woman will attest….. do not get up and drink water in the morning, eat a soda cracker or two. You are simply [suffering] a little nausea.