 Ozone (O3), a triatomic form of the ordinarily diatomic oxygen (O2), is a curious species. We will discuss some of ozone’s curious physical properties. We will not include information related to health or the environment.
Ozone (O3), a triatomic form of the ordinarily diatomic oxygen (O2), is a curious species. We will discuss some of ozone’s curious physical properties. We will not include information related to health or the environment.
Molecular Bonding
Just as it takes two points to determine a straight line, diatomic oxygen is also linear. It’s bond length is 1.208 Å (equal to 120.8 pm).
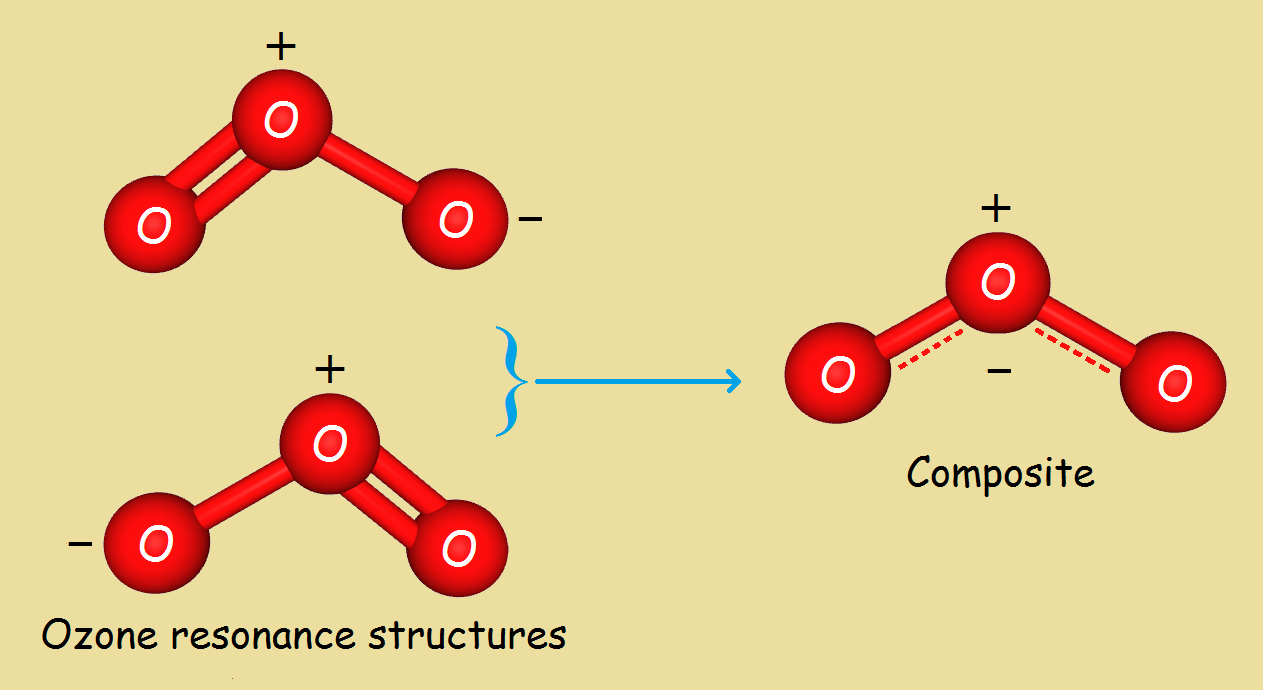
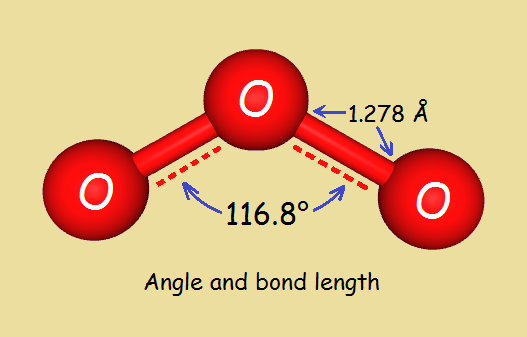
Ozone is not linear; it is bent at an angle of 116.8o, which is greater than the measure of the angle for a water molecule, 104.5o.
Comparing Boiling Points
The third oxygen atom plus the relatively large angle suggests ozone should boil at a higher temperature than oxygen. In fact, this is the case.
This is because the bent ozone molecules, as seen in the illustration, produces a distribution of charges, plus and minus. This strengthens intermolecular forces, which might be loosely compared to the hydrogen bonding between water molecules.
Just as hydrogen sulfide boils at a temperature some 60oC below the temperature of water due to hydrogen bonding, oxygen boils at a temperature 71oC below that for ozone. Ozone boils at -112oC, while oxygen boils at -183oC.
The Liquid State

It is not only the boiling points that are different. Liquid oxygen is strongly paramagnetic, thanks to having two unpaired electrons, whereas liquid ozone is diamagnetic, having all its electrons paired.
Although liquid O2 is light blue in color, liquid O3 is deep indigo in color, much more intense.
The Solid State
As might be anticipated, solid oxygen is light blue, like the liquid. However, we here embed a most lovely video that demonstrates the forming of solid oxygen in the laboratory (2:57).
What about solid ozone? It darkens yet further as the indigo liquid freezes into a deep blue-black solid.
Magnetic Properties
This, our second video, explains paramagnetism and diamagnetism. Note especially the first half of the video (about 4 minutes).
Note: You might also enjoy Hydrogen Metal?
References:
