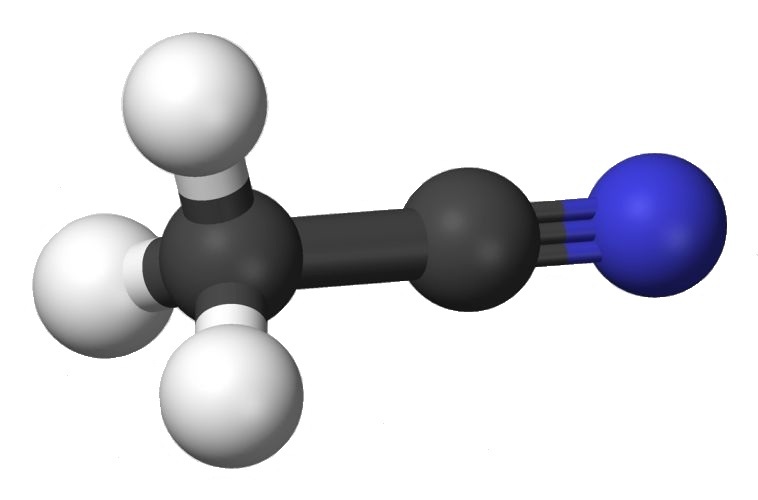
 Hydrolysis of acetonitrile (the adding of water to its triple bond) is a useful synthetic procedure.
Hydrolysis of acetonitrile (the adding of water to its triple bond) is a useful synthetic procedure.
Alkali metals such as sodium (Na) and potassium (K) attached to the cyanide group (carbon and nitrogen joined by three bonds, ‒C≡N or -CN for short) is deadly poisonous.
One factor making it so poisonous is most inorganic cyanides ionize in water. Dissolving cyanides separates the cation (for example, Na⁺) from the cyanide ion, ‒CN⁻.
When an organic group is attached to ‒CN, the group is not ionized, even if the organic is water soluble. In such a case, the compound is called a nitrile. Nitriles are generally much safer to handle.*
What some might call methyl cyanide, CH₃CN, is more accurately acetonitrile. Why acetonitrile? Hydrolysis of the ‒CN group produces acetic acid.
Hydrolysis of Acetonitrile
The simplified reaction is,
CH₃CN + 2 H₂O → CH₃COOH + NH₃
The hydrolysis of acetonitrile essentially involves adding water across the triple bond.

The Steps of the Hydrolysis
In actuality, the mechanism of the hydrolysis of nitrile differs, depending upon whether one uses an alkaline or an acidic hydrolysis. The mechanism of the alkaline hydrolysis is seen in this Boise State University (Idaho) schematic.
The acidic hydrolysis is seen in this schema, also by Richard C. Banks of Boise State University.
Hence the Name
For the name acetonitrile to be obviously relevant, the reverse procedure should be achievable. It should be possible to form acetonitrile from acetic acid. Is that the case? It is, though some might find it just a little bit complex and not practical for laboratory preparation.
Acetonitrile from Acetic Acid
Consider this article from Tomsk State University (Томский государственный университет), Siberia Russia. It informs us that carefully controlled conditions and an appropriate catalyst may be used to convert ammonia and acetic acid (with a few other minor additives) into acetonitrile.
*This is not to suggest it is safe to handle nitriles irresponsibly.
Note: You might also enjoy Acetylene and Acetylides
References:
- Princeton University: Acetonitrile
- Wichita State University: Acrylonitrile, hydrogen cyanide, and crude acetonitrile
← Back to Classic Science
← Home
