 Although individuals and governments fall far short of maintaining Earth’s pristine qualities, many do desire to minimize waste and its first cousin, pollution. Although one admirable action would be to minimize production, another is to recycle the resultant wastes.
Although individuals and governments fall far short of maintaining Earth’s pristine qualities, many do desire to minimize waste and its first cousin, pollution. Although one admirable action would be to minimize production, another is to recycle the resultant wastes.
The late 19th and early 20th centuries introduced industrialization, and with it mechanization. Part of the mechanization was the automobile. And part of the automobile was the automobile tire. Tires are constructed of rubber or rubber-related products. They wear out well before most automobiles wear out.
The problem is: what to do with all those worn-out, used tires? Millions and millions of them. They do not readily break down on being exposed to the elements. How can they be recycled?
One Problem with Tires Now
What might happen if tires were buried in a landfill? Well check this video out to see!
This reminds me of the underground fires burning in some of Pennsylvania’s former coal mines. This one has been burning for decades…
Recycling
There are creative uses for old tires. Some percentage of them are “recapped” with fresh rubber for continued use. Others are converted for use on playgrounds. Some are converted into floor mats. They’ve even been modified and painted white and used as outdoor flower containers!
Vacuum Distillation
For a couple/few decades, scientists have investigated whether it is practical to convert tires into limonene, which is. chemically, a terpene. After all, tires are derived from isoprene, the simplest of the terpenes. The major component of the finished tires is cis-polyisoprene.
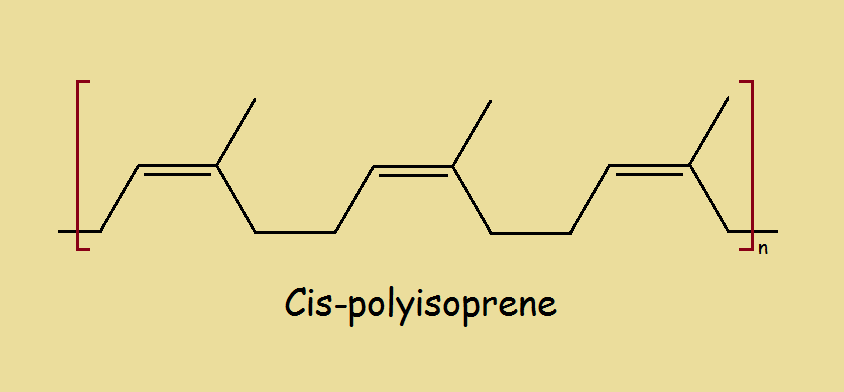
Commercial feasibility is dependent, in part, on yield. How much limonene can be prepared per pound of tires? Maximum yield occurs at temperatures somewhere between 400o and 500o Centigrade. Temperatures above 500o crack some of the limonene molecules, decreasing yield.
Additives
Certain additives are said to increase yield. Will an additive be discovered, along with finely tuned parameters, that will favor recycling tires in this fashion? Time will reveal “the rest of the story”.
Note: You might also enjoy Algae – When Life Hands You a Lemon
References:
- Journal of Analytical and Applied Pyrolysis: Production of dl-limonene by vacuum pyrolysis of used tires, by H Pakdel, et. al. (2001) [PDF]
- Journal of Analytical and Applied Pyrolysis: A review of dipentene (dl-limonene) production from waste tire pyrolysis, by B Danon, et. al. (2015)
- Discover Magazine: Lemon Tire Very Pretty… (1997)

Hmm, I am not sure I would enjoy using lemon oil if I knew it came from recycled tires. Some tires are being used in building: filled with soil and used as walls. Some tires are shredded and used to create non slip road surfaces.