 Many enjoy hot asparagus, whether as a cream soup, or a simple steamed, sautéed, or even raw vegetable. Since I am a chemist, I find it of interest from that perspective as well. Yes, I realize the reader almost certainly has already deduced asparagusic acid is named for this grass-related vegetable.
Many enjoy hot asparagus, whether as a cream soup, or a simple steamed, sautéed, or even raw vegetable. Since I am a chemist, I find it of interest from that perspective as well. Yes, I realize the reader almost certainly has already deduced asparagusic acid is named for this grass-related vegetable.
Background
But let me give a little background before I get into asparagus and asparagusic acid, and why I write concerning them.
When I was the age for it, I attended Drexel Institute of Technology1 in Philadelphia, PA. Drexel offered the benefit of a cooperative work assignment each year to provide job-related experience plus a small income to help cover the cost of college.
My first assignment provided an additional perquisite: a government car for transportation to Trenton, NJ. I was not aware the vehicle was for my exclusive use, so my supervisor set it up that I would be dropped off at his house and he would drive.2 Not a terribly ethical fellow.
His Son
The boss had a son whom I rarely saw, and only at a distance. He was in his 20s and drove an open-air, sporty Jaguar automobile, undoubtedly an older model. It became apparent my boss had mixed feelings concerning his son.
Although he didn’t approve of everything his son did, he seemed to appreciate the sneakiness his son displayed in the event I will now relate. Did you guess it concerns asparagus?
The Son Receives an Invitation
My boss informed me
“My son wanted to dodge the draft, so he hatched out a scheme. He ate tons and tons of asparagus and acted crazy during the pre-enlistment procedure.”
Sadly, I don’t recall for a certainty whether he succeeded in avoiding military service or not. Certainly if he was successfully drafted, it was not for the son’s lack of trying to avoid it!
But Why Asparagus?
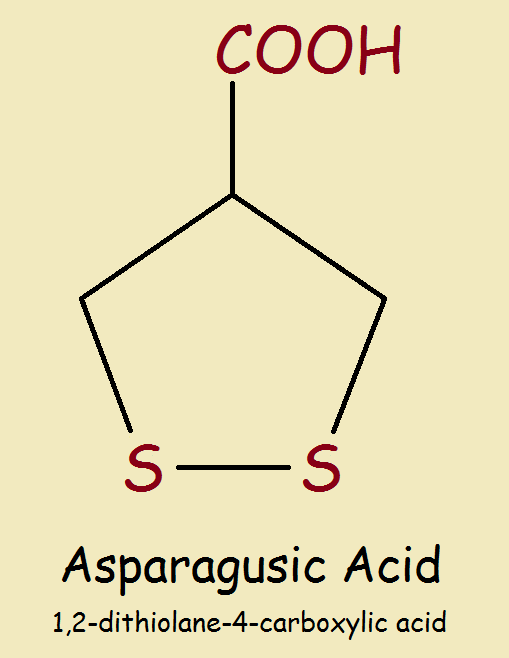
Asparagus is pretty much unique in its containing asparagusic acid. Notice the chemical structure in the image provided. The 5-member ring consists of 3 carbon atoms (C) and 2 sulfur atoms (S). Carbon atoms are often not represented by C, which is understood.
The group at top, or -COOH, is the carboxylic acid group. This group is common in food, as for example, in vinegar or acetic acid, CH3COOH.
Elemental sulfur is a nearly odorless solid. But when sulfur occurs in volatile compounds, it carries with it a distinct odor. When asparagus, and hence, asparagusic acid, is consumed, it breaks down in the body producing wastes or metabolites.
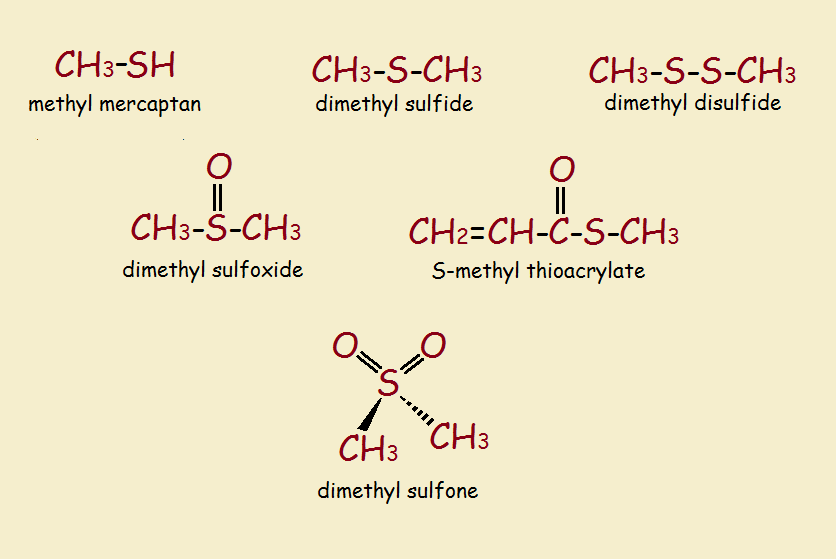
Asparagusic acid apparently is responsible for the production of a whole host of sulfur-containing metabolites. See the second image for the names and chemical structures of some of these.
Presumably, then, my supervisor’s son counted on receiving a thumbs-down urine test or general diagnosis that would disqualify his serving. As I say, I don’t recall whether or not his effort resulted in a successful venture.
1 Now Drexel University
2 I learned of this later thanks to a co-worker who squealed on him.
3 Not his exact words
Note: You might also enjoy Yum! Cicada Stir Fry
