
Definition: azeotropes are a specific mixture of compounds that boil together as if they were a single substance, at constant temperature.
Individual Boiling Points
In most instances, liquids boil as pure substances at a specific temperature. It is this property that allows boiling (as a physical property) to be used as a means of separation. For instance, an ester may be produced by reacting an organic acid and an alcohol. The resultant ester may usually be separated by boiling from excess alcohol or excess acid.
Yet, Occasionally
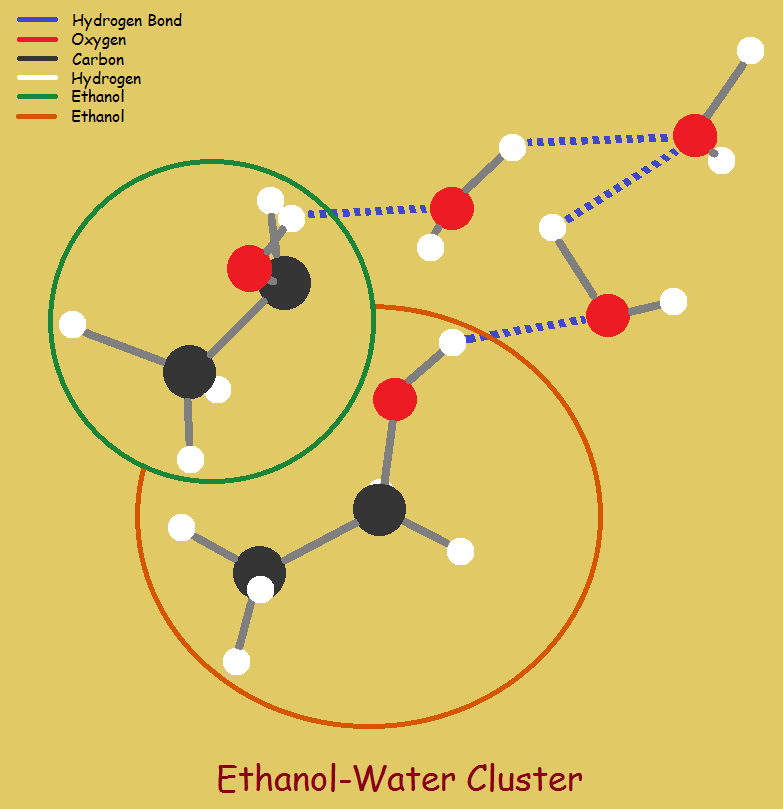
In some instances, however, as for example in the case of ethyl alcohol and water, there may be a mixture that distills over, instead of a pure substance. Thus a “mixture” of approximately 95.6% alcohol and 4.4% water is distilled over before either pure alcohol or pure water is left to distill.
Azeotropes – Things Unseen
Now that we know what an azeotrope is, why do they exist? It has to do with stability. Stability in the vapor phase. I am inclined to think it is similar to the process of dissolving a crystal of solid such as sodium chloride (NaCl) in water. Molecules of the NaCl are bonded to water.
A polar molecule can be surrounded by and bonded to a number of molecules of water. By the same token, I believe a molecule of distillate can be surrounded by one or more molecules of co-distillate.
For ethanol and water I would not be surprised to learn a molecule of water is encircled by “x” molecules of the ethanol. Energy is at a minimum; stability is at a maximum. All of this may already have been proven or disproven. I do not know.
Whatever the case, I think it makes sense, so I share it with you.
Note: You might also enjoy Chemical Separation by Fractional Distillation and Crystallization
References:
← Back to Classic Science
← Home

Is this process similar to adding fuel additive to a tank of gasoline in an automobile to remove water from the gasoline?
Although my answer must be, “Not exactly,” at the same time I must say, “In a sense.” If an ingredient is added to remove the water from an azeotrope, one component of which is water, the answer must be, “Yes.”