 Curiously, the aroma chemicals in coffee closely resemble the stench-producing chemicals in the abominable skunk spray! They are similar, but they are not identical, and they do not occur in proportionate quantities.
Curiously, the aroma chemicals in coffee closely resemble the stench-producing chemicals in the abominable skunk spray! They are similar, but they are not identical, and they do not occur in proportionate quantities.
Skunk Stench
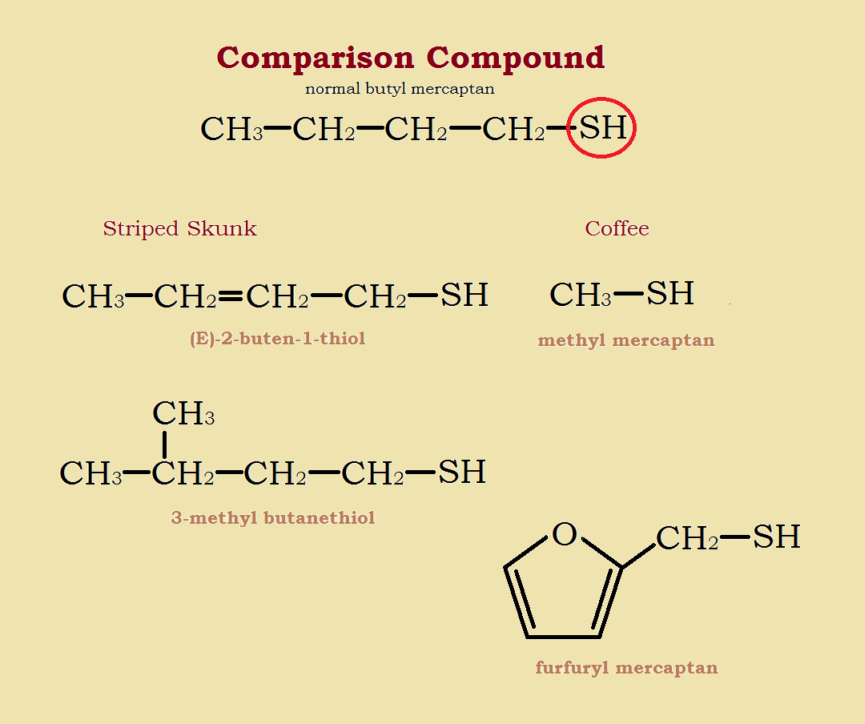
The chemicals that primarily give skunk spray its stench are thiol derivatives—in particular, derivatives of the sulfur alcohol n-butyl mercaptan (CH3-CH2-CH2-CH2-SH).
The portion of this compound responsible for its smell is the -SH group, a sulfur atom bonded to a hydrogen atom, which is similar to the -OH or alcohol group. Common derivatives modify the carbon chain. Another variation chemically modifies the -SH group, using acetic acid (CH3-COOH) to form the corresponding thioacetate.

Two of the most odoriferous skunk spray mercaptans are (E)-2-buten-1-thiol and 3-methyl-butanethiol. Other than its prevalence (it makes up approximately 2/5 of skunk spray) a factor that could intensify its aroma is its double bond, which makes it an alkene mercaptan. The basic carbon chain is not CH3-CH2-CH2-CH2–, but CH3-CH=CH-CH2–. Similarly, the brown marmorated stinkbug produces two foul smelling compounds; both are aldehydes and alkenes.
Coffee Aroma
There are two mercaptans (thiols) especially responsible for coffee aroma—methyl mercaptan (CH₃-SH) and furfuryl mercaptan (C4H3O-CH2-SH). Typically, the smaller a mercaptan molecule is, the more volatile it is, and the more intense its aroma is likely to be. However, these mercaptans are present in very small amounts in coffee. It takes a gas chromatograph to detect them!
In Conclusion
So when is it that the presence of a skunk is likely to smell most like our delicious coffee aroma? It is long after the initial presence of our striped or spotted friend—when its mercaptan levels have diminished, causing its odor to have faded considerably.
Note: You might also enjoy Why Dogs Pee on Tires
References:
- Chemistry of Skunk Spray
- Alarm Odor Compounds of the Brown Marmorated Stink Bug…
- Flavor Scientist: Coffee and Furfuryl mercaptan FEMA 2493

Okay, now that I have read this, I will go have a cup of coffee later on in the day but try to make it stronger so I can get more punch from the aroma. LOL
I’ve smelled sites where skunks have been (where the aroma is not too fresh or too strong), and they really do smell like fine coffee!
We have skunks in my neighbourhood. Many times I’ll wake up, thinking I smell coffee, then realize it’s actually skunk. Funny how something so good can almost smell like something so bad!
You’ve got that right!
Fresh brewed coffee, all coffee, smells like fresh skunk musk to me. I can’t stand coffee because of this.
You mean it didn’t make you want to snuggle-up with a skunk because skunks smell so good? LOL.
I am a coffee-holic and love the aroma. I’ve wondered for a long time if the chemicals in coffee and skunk spray have similarities, because they smell similar to me sometimes. Oddly, faint skunk scent doesn’t offend me as much as it does most people… now I know why!
Same here. I’ve gotten so if I’m driving through an area of isolated businesses and the odor wafts past me I wonder, Is this coffee or is this skunk?
“Funny” anecdote, I had an old Dodge van for about 3-4 years, in the mornings it was fine, but after a 5 or 10 mile drive, there was a definite skunk smell! And the odor was still there when I gave it away. For a long time, I thought the smell was coming from under the house, but finally figured it out. Buuut…. I just wondered if possibly that might be the reason the Chevy dealer gave me such a good deal?!?!
Furfuryl mercaptan does not have the -CH2SH group bonded to the oxygen. It’s bonded to the carbon next to the oxygen.
Quite right. I must have missed a cup of coffee that morning! Thanks for the good eye. Fixed.
I have been meaning to look this up for years. Finally got around to today and it’s good to know my nose is correct. I wonder if this similarity only occurs to black coffee drinkers like me? It seems that the concoctions that are served at Starbucks might not qualify.
I used to love coffee and the aroma, until recently when I realized that I can no longer drink caffeine. It’s been about 10 months since and now all roasted coffee smells like skunk. It is so weird, some mornings I start to worry, thinking my dogs got sprayed by a skunk when it was just a family member brewing a cup of Jo…
Really?
Skunk “stench”?
“abominable skunk spray”?
Don’t these beautiful creatures have enough problems without pejorative language sprayed on them?
But yeah, I had not made the connection. I drink lots of coffee, and some years ago discovered that I actually enjoy the fragrance of skunk scent, in limited quantities.
It is always possible to have too much of a good thing.
Heh-heh. The spray, not the critter. They’re actually rather cool, though I believe I’d rather have a raccoon for a pet.
Someone at our office just walked by my desk with the skunkiest coffee I’ve ever encountered. It’s so pungent that it’s becoming nauseating. So I started searching for an explanation of this smell, and that’s how i ended up here. Thanks for the info!
My pleasure.
Thank you for validating my olfactory senses! My husband has always cited my belief in the similarity of skunk odor and coffee as a symptom of insanity! Haha! I win!
Hands down!
I love the scent of skunks, not sure why people complain about it. It’s such a a delicate aroma. Oh I also like the smell of coffee.
What about skunk-smelling armpits after drinking coffee? I know two related people who experience this.