 Potassium and sodium cyanides are iconic poisons. Murder mystery movies frequently feature one of these two substances as the cause of death.
Potassium and sodium cyanides are iconic poisons. Murder mystery movies frequently feature one of these two substances as the cause of death.
Their chemical formulas may be written more than one way. Most simplistically, they are KCN and NaCN. K stands for potassium (kalium), and Na stands for sodium (natrium). C is for carbon. N is for nitrogen.
Another way of writing the chemical formulas of these substances is, K–C≡N and Na–C≡N. Both of these chemical compounds dissolve in water. This makes them especially dangerous, since if consumed, they can enter the bloodstream and travel to all of the bodies’ cells.
KCN → K+ + CN–
NaCN → Na+ + CN–
Cyanides How Toxic? Organic Nitriles
Organic nitriles are a kind of cyanide. For instance, acetonitrile is written,
CH3CN
But this looks like methyl cyanide! It is, in a sense. Yet again, it is not. A true cyanide yields the CN group in ion form. That is, it produces a cyanide ion, CN–. Although acetonitrile dissolves in water, it does not break apart into ions. Entire molecules dissolve, remaining undivided. Take special note of that fact!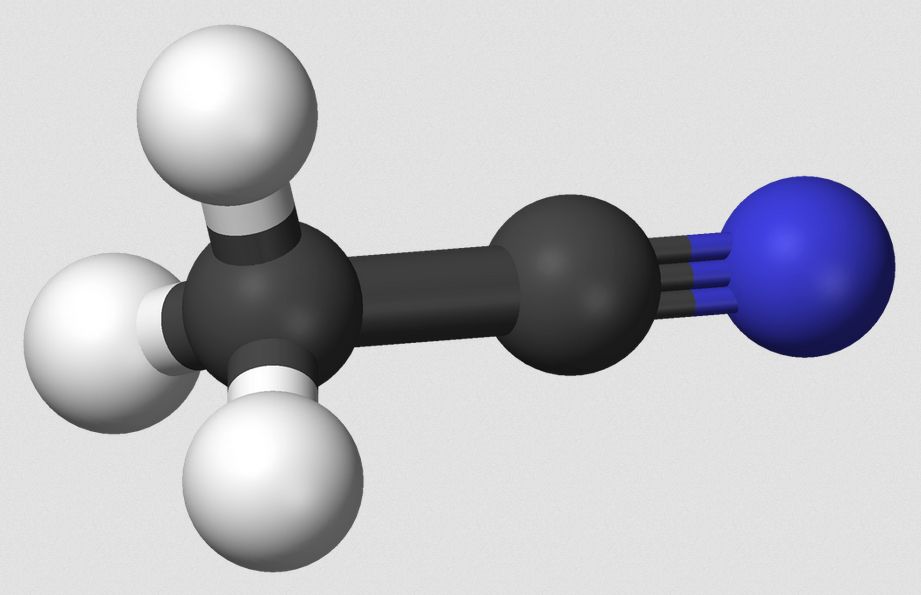
Such compounds are toxic if metabolized1 to be sure, but they are not nearly as toxic as water soluble, ionizable cyanides.
Cyanides How Toxic? Polymeric Nitriles
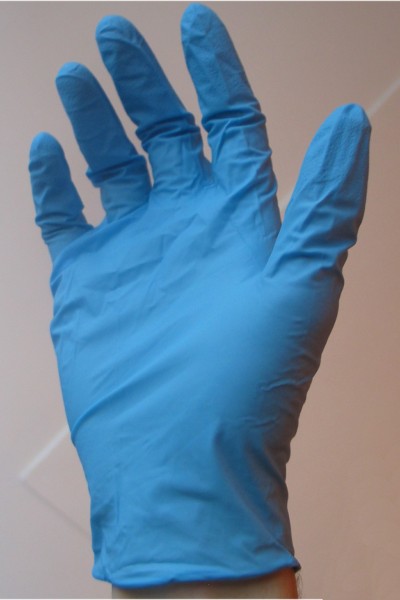
Visit your physician, and in his or her waiting room you are likely to notice boxes of rubber gloves. In particular, you may notice some of these gloves consist of nitrile rubber. These gloves are made of insoluble rubber that contains attached nitrile groups. They pose no health threat to the patient or physician or his or her staff. This “cyanide” is perfectly and completely harmless.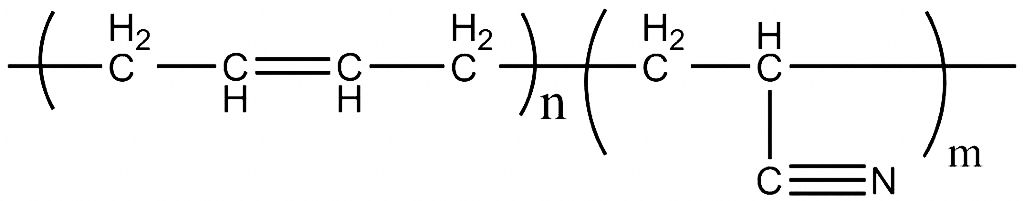
I had the great pleasure of pointing this out to my own family physician, namely that the gloves he wore were made of a polymer containing cyanide groups. Being an intelligent man, he took it philosophically, not being alarmed at all.
1 metabolized refers to a breaking down by the body, which in this case, takes hours.
Note: You might also enjoy:The Hydrolysis of Acetonitrile or Methyl Cyanide
References:

It’s amazing how different combinations of the same substances have such different properties!
Question not answered. Are insoluble inorganic cyanides as deadly as those that dissolve in water? Example: Is insoluble Aluminum Cyanide as deadly as Sodium Cyanide. Does it affect the nose and throat if inhaled like NaCN? I have made 30 different inorganic cyanides that are insoluble in water.
All cyanides are toxic to some degree. Insoluble compounds are essentially never completely insoluble. Not 100%. Some compounds are noted to be very, very close to completely insoluble. For instance, barium sulfate. The medical industry depends upon its near perfect insolubility. But most insoluble compounds dissolve to at least some extent.