When two different types of atom are bonded together, they do not share their bond electrons equally. This is because each type of atom possesses its own charge environment, which results in an atom’s electronegativity.
Electronegativity is the measure of an atom’s ability to attract additional electron density to itself.
For example,
Sodium seeks to give an electron to become a positive ion, Na+. It has a very low electronegativity.
Iodine wants to gain an electron to become a negative ion, I–. It has a relatively high electronegativity.
Charge Environment
Atoms vary in electronegativity, and bonds vary according to constituent atom electronegativities. The electronegativity of an atom depends upon its charge environment. That environment depends primarily on three things…
- Distance between charges,
- Total charge and
- Shielding effects
Distance Effect and Attraction
Positive and negative charges exert attractive force on each other that varies as the square of the distance between them. If an electron and a proton are doubled in distance from each other, the attraction decreases by a factor of four. If they are quadrupled in distance from each other, the attraction diminishes to one-sixteenth what it was.
It’s Elemental: Total Charge
Elements differ in the number of protons in their nuclei. Hydrogen has 1, helium 2, lithium 3, oxygen 8, bromine 35 and gold 79. It might seem that this should be the primary determinant of electronegativity, but it’s not.
To illustrate: Fluorine has a mere 9 protons, while sodium has 11. Yet sodium has a very low electronegativity, while fluorine has the highest electronegativity of all the elements. The greater the number of its protons, the larger the nucleus is and the larger the atom is.
Increasing atomic size gradually decreases electronegativity.
The following video, for the first 2 minutes and 15 seconds or so, crystallizes these concepts exceptionally well.
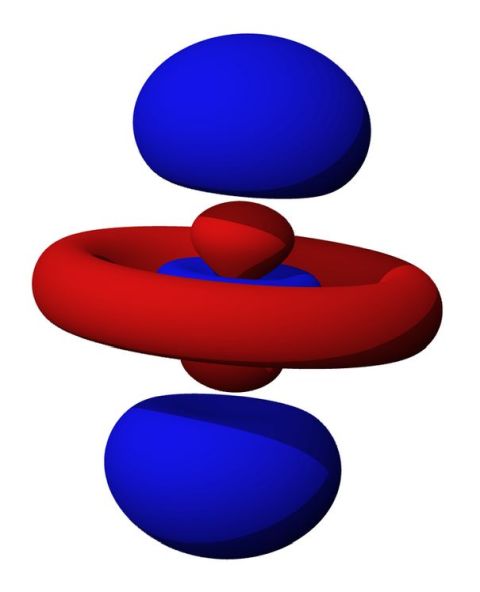
Shielding Effect
An increase in electrons in an atom forces some of the electrons to occupy different orbital types; the larger the atom, the more complex the orbitals become. Helium has only s-orbital electrons, with simple spherical symmetry. Carbon has s- and p-orbital electrons. P-orbitals are more complex, shaped something like dumbbells. Strontium has s-, p- and d-orbitals. The reader will note just how complex the shape of a 5d orbital is from the image above. Radon has s-, p-, d- and f-orbitals.
Complex Orbitals and Electron Density
Complex orbital types alter the geometrical distribution of electron density or electron richness surrounding the nucleus. Inner electrons shield outer electrons, and increased shielding decreases electronegativity. This is because the outermost electrons do not feel the force of the positive charges of the nucleus as strongly. Those electrons thus reside a greater distance from the nucleus and are held less strongly. There is also little tendency for the atom to draw in additional electrons.
If shielding is decreased, the opposite occurs. Outer electrons are drawn in more closely and the atom is more capable of accepting additional electron density… Then, the atom has greater electronegativity.
Note: You might also enjoy Calculate Atom Weight Two Ways
References:
- University of Nebraska. Polarity of Bonds and Molecules
- MM’s PhysChem. Electronegativity vs. Atomic Number
- Virginia Tech. Electronegativity and Polar Bonds
