 Teflon® is famous for its smooth surface. Essentially nothing sticks to Teflon. It has been bragged that the only thing that can stick to it may be a gecko.
Teflon® is famous for its smooth surface. Essentially nothing sticks to Teflon. It has been bragged that the only thing that can stick to it may be a gecko.
This is because of the special nature of a gecko’s grip. In fact, it appears a gecko can only stick to Teflon under special conditions. So the question is: How can we glue Teflon?
I Need to Know How to Glue Teflon
And yet, that was the task I was given at my place of employment. I was told the use of a special chemical pre-treatment could allow objects to be glued to the slippery stuff. Since I am a chemist, the reader can rest assured this piqued my interest immensely. What was the pre-treatment, and how did it work? The manufacture of the stuff was not about to reveal it to me. It was proprietary!
I Had to Know, But Not Tell
Out of respect for the manufacturer, I won’t mention the name of the product. But I will tell you what I deduced about it, and why it is logical I drew all the correct conclusions. First, the stuff was a sodium metal reagent mixed in with tetrahydrofuran (THF). This is a solvent much like diethyl ether. Having specialized in organic chemistry, I realized it was an organometallic chemical reaction.

Upon combining the reagent with water, one notes the distinct odor of moth crystals. Not paradichlorobenzene, but naphthalene. This is a simple two-ring aromatic hydrocarbon, chemical formula C₁₀H₈. If this is the organic “principle” and it was combined with sodium to form an organometallic compound, then likely the compound had one of those 8 hydrogen atoms replaced by a sodium metal atom.
Am I Just Crazy?
Was I crazy? Could such a compound exist? It does! The online Science Direct website cites Tetrahedron Letters, Volume 12, Issue 47, Pages 4581-4583, which speaks of the reaction of the Grignard (organometallic) reaction of sodium naphthalene with organic halides. Why, the solvent of choice is even tetrahydrofuran!
But is Teflon an organic halide? Absolutely it is! The halogen involved is fluorine. Teflon, after all, is nothing more than polytetrafluoroethylene. The structure basically consists of a chain of carbon atoms whose pendant or attached atoms are atoms of fluorine.
How to Glue Teflon?
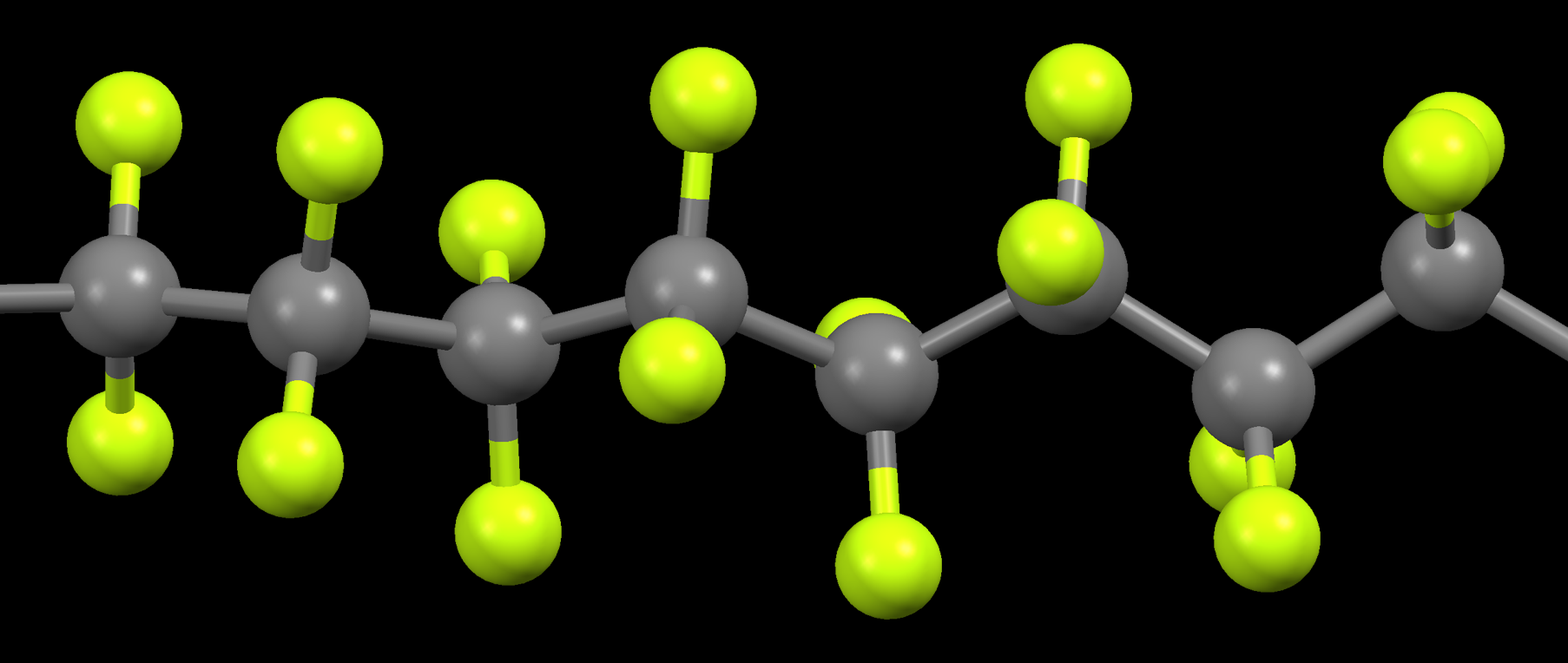
Combine the reagent with the portion of the Teflon to be glued, and the fluorine atoms react with the sodium metal to form sodium fluoride, while the naphthalene attaches to the outermost layer(s) of the Teflon. It is actually those chemically bonded naphthalene rings one glues to. This outermost layer is very thin, so the bulk of the Teflon maintains it characteristic properties.
All of this bespeaks a successful inference, but is there more evidence this is the reagent that was applied to enable Teflon to be glued? Yes, there is just a little more. It has to do with the color of the Grignard reagent itself, the manufactured product. What color is that? It is a very deep, dark, blue-green. How does that prove anything? Sodium naphthalenide’s deep green color arises from spectral absorptions centered at 463 and at 735 nanometers wavelength. Yes, the color is the same!
So What Enables It?
So, in conclusion, there is as yet no known glue—no adhesive—that can successfully be applied directly to Teflon and result in a firm bond. It is only by means of chemically modifying a portion of the molecule so its chemical makeup is no longer strictly Teflon, that gluing can be accomplished.
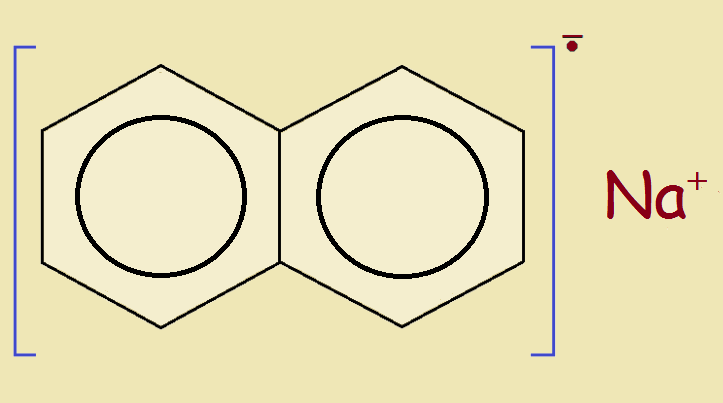 An organometallic product that modifies the structure to replace some of the fluorine atoms with naphthalene molecules has successfully allowed adhesion to objects made of Teflon. This was once used in connection with a device designed by electrical engineer, Anthony R. Kerr, a man I knew and worked with in the field of radio astronomy.
An organometallic product that modifies the structure to replace some of the fluorine atoms with naphthalene molecules has successfully allowed adhesion to objects made of Teflon. This was once used in connection with a device designed by electrical engineer, Anthony R. Kerr, a man I knew and worked with in the field of radio astronomy.
Note: You might also be interested in Anodizing Aluminum: How Can It Be Done?
References:
- U. of Akron: Gecko-Inspired Adhesives & Tapes
- Organic Syntheses: Org. Synth. 1987, 65, 166
- NRAO – Charlottesville, VA: Anthony R. Kerr
- N. G. Connelly and W. E. Geiger, “Chemical Redox Agents for Organometallic Chemistry”, Chem. Rev.

That is so damn interesting!!!!!!!!!!!! I LOVE ORGANIC CHEMISTRY!!!!!!!!!!!!!! THANK YOU!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
You can use Polytetrafluoroethylene (PTFE)
Yes, this is the same as Teflon, so the technique does work for it.
Thanks for the information.
Is there any adhesive which you test for teflon after chemical treatment that works great?
I’m not quite certain what you mean, but I *think* you mean is there some specific adhesive to use to “glue” treated Teflon? Well, the treated Teflon has a thin coating of naphthalene molecules that you “glue to”. Whatever works as an adhesive for naphthalene should work well on the treated Teflon. Keeping in mind the naphthalene layer is thin, doubtless judicious application is called for.