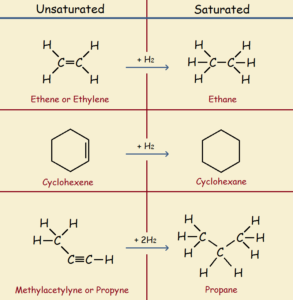
Hydrogenation is simply the addition of one or more molecules of hydrogen to a compound.
Hydrogenation is simply the addition of one or more molecules of hydrogen to a compound.
When we speak of hydrogenation, we usually refer to the saturation (adding of hydrogen) of carbon-carbon double bonds to produce single bonds.
Saturation / Unsaturation
Organic compounds, most of the compounds containing carbon and hydrogen, can be saturated or unsaturated. In saturated compounds, all carbon atoms that are bonded to other carbon atoms are singly bonded, whereas in unsaturated compounds, some of these are double or even triple bonds. See the illustration for examples.
But Why Hydrogenation?
Whatever the actual motive(s) involved, the medical and commercial worlds decided butter should be replaced by a manmade product, initially called oleomargarine, and later margarine.
In order to be “healthy” we are told it should be prepared from vegetable oils rather than animal fats. The process of hydrogenation would be used to convert the liquid vegetable oils into spreadable solids.
Melting Point Comparisons
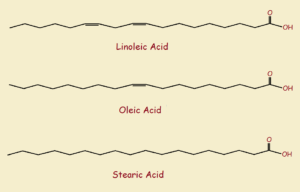
Does hydrogenation effectively do that? It does. Due to the complexity of the composition of the various vegetable oils, we will consider only two of the major component fatty acids1 found in vegetable oil – linoleic and oleic acids. Both of these organic acids have an 18-carbon straight-chain structure.
Upon hydrogenation, the pure saturated acid resulting is, in both cases, stearic acid. Stearic acid melts at a temperature of 156.7°F or 69.3°C. What is the melting point of pure linoleic acid? 23°F or -5°C. And oleic? 56°F or 13.5°C.
Butter and Margarine Updates
Eventually, as is so often the case,2 there was at least a partial return to butter.
Not all have returned to its use, however. Some have come to prefer the flavor or consistency of a particular margarine they’ve grown accustomed to.
Others have favored the use of margarine for certain cooking applications. If careful watch is not made, butter is famous for burning. Partly for this reason, some embrace the combining of butter with olive oil – olive oil for its unsaturation and cooking properties, butter for its taste.
Final Remarks
No comments about the relative merits of margarine or of butter will be made here. First, there is a lack of medical qualification on the author’s part. But more importantly, he has become all too well aware how changing the “experts” can be when it comes to evaluating the merits of one food over another.
1 It should be noted that one molecule of fat is made up of one molecule of a fatty acid and a molecule of glycerin. Thus a saturated fat is made up of glycerin and a saturated fatty acid. An unsaturated fat is made up of glycerin and an unsaturated fatty acid.
2 Take for example, the substitution of carob for chocolate. Carob was thought to be a healthy substitute for the delinquent chocolate.
Note: You might also enjoy What are Cis and Trans Double Bonds? That’s Easy
References:
← Back to Food and Health
← Home