 How are nitrogen heterocycles formed from aromatic amines?
How are nitrogen heterocycles formed from aromatic amines?
Aliphatic hydrocarbons¹ take a number of forms. The simplest is written CH₃(CH₂)nCH₃, where n is a positive integer.² Replace a hydrogen atom at one end of the chain with a carboxylic acid group (-COOH) and a hydrogen atom at the other end with an amino group (-NH₂), and you have a straight chain amino acid.
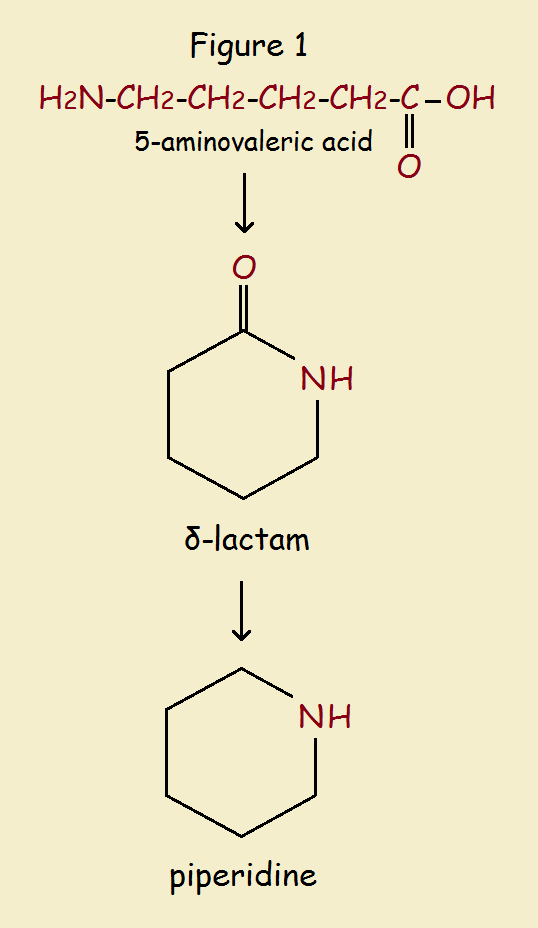
For instance, choose n = 3. That gives the molecule
HOOC-CH₂-CH₂-CH₂-CH₂-NH₂
Its name is 5-aminopentanoic acid or 5-aminovaleric acid.
 Cyclization
Cyclization
Under the right conditions, this species can be cyclized to form an amine salt. The salt is then converted to a cyclic amide, a delta lactam. The lactam is next reduced to the amine piperidine. The overall process is illustrated in Fig. 1.
Nitrogen Heterocycles
A cyclic amine, a compound with nitrogen in the ring, is a nitrogen heterocycle. The prefix hetero- means “other.” Nitrogen “another” non-carbon atom. If the nitrogen is not within the ring, but is merely atttached to the ring, it is not a heterocycle. So aminocyclohexane, a six carbon atom ring with an attached amino group, is not a heterocycle.
 Additional Examples
Additional Examples
Another example is 2-methylpyrrolidine. It is prepared from 3-methyl-3-aminobutyric acid. The reaction is illustrated in Fig. 2.
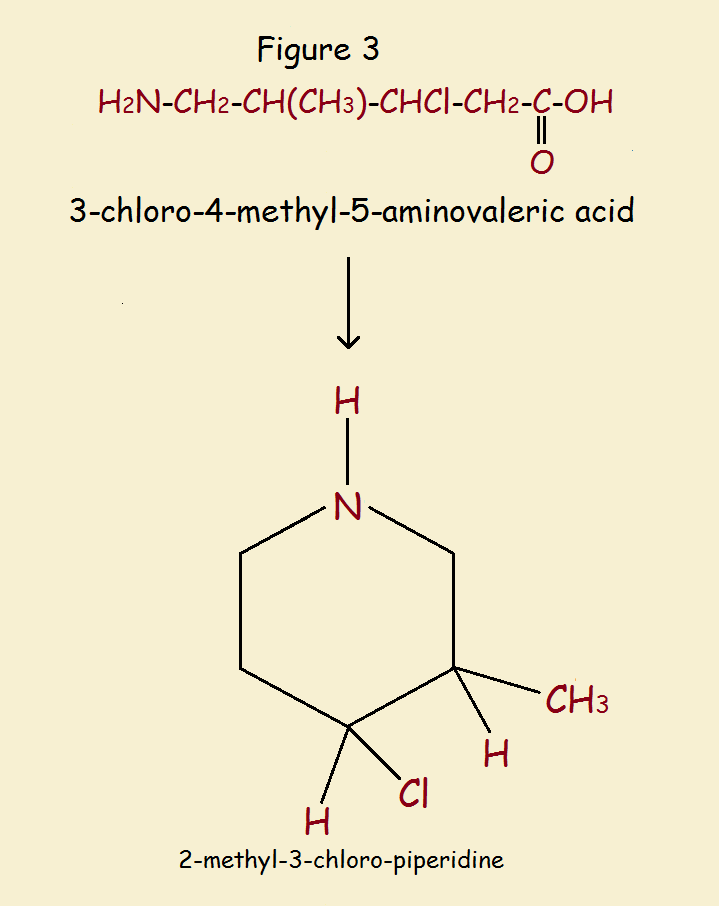
A final example is the cyclization and reduction of 3-chloro-4-methyl-5-aminovaleric acid to form 2-methyl-3-chloro-piperidine. See Figure 3.
¹ An aliphatic compound is an organic compound characterized by an open chain, not rings.
² The letter C stands for carbon; the letter H stands for hydrogen.
Note: You might also enjoy DNA: The Modular Staircase of Life
References:
← Back to Classic Science
← Home

I ought to include an explanation of the amino acid becoming an amine salt. At that step, the acid -COOH group combines loosely with the amino group -NH2, to give -COO with a minus charge and -NH3 with a plus charge. Heating that releases water, producing the group -CONH- which is like the clasp on a string of beads. That structure, upon reduction, produces -CH2-NH- (in other words, the heterocyclic amine structure).