Scientists desire to solve complex problems with exact precision, but sometimes it just is not practical. Simplifying is necessary. For the organic chemist, one form of simplifying is the idea of pushing electrons.
To illustrate, high school physics instructors introduce the concept of massless strings and frictionless pulleys. No such things exist. Still, this fiction enables the beginning student to isolate what is important. Spark Notes informs us that college entrance examinations generally employ such contrivances.
Constructs, Artifices, Contrivances
Physicists are not the only ones to employ constructs and contrivances to simplify problems and arrive at an answer. The organic chemist must understand very complex compounds and the reactions leading to their formation.
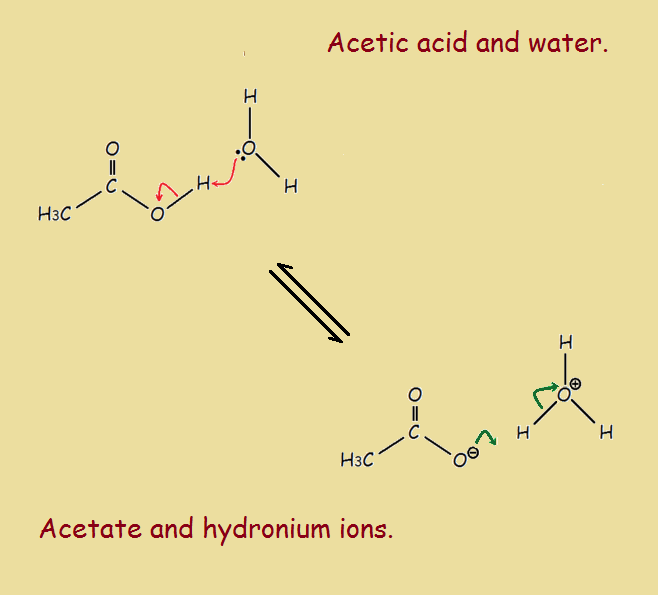
One of the best known contrivances is that of pushing electrons or pushing arrows. The great thing about pushing electrons is that it does not lead to mere approximation, but produces accurate answers. What is electron pushing and how does it work?
Simply put, pushing electrons refers to the transfer of electrons from the filled orbital of one bonded atom into another unfilled orbital, whether a p-orbital or an anti-bonding orbital of another bonded atom, whether a pi or a sigma orbital. This latter action breaks a formerly existing bond. In fact, there is usually a sequence of such conversions during a chemical reaction.
The Art Behind Pushing Electrons
The pushing is illustrated by the drawing of a curved or ‘curly’ arrow. There may be many such arrows drawn in the illustration of a reaction mechanism, but it is usually wisest to list a separate drawing for each individual transition of the mechanism.
Enjoy this fantastic video on the subject, by the University of California – Irvine. It is both very thorough and authoritative.
Note: You might also enjoy The Simple Difference Between Alkanes, Alkenes, and Alkynes
References:

Oh! I never heard of electron pushing before. It must be a new way to explain how things work. Although, of course when I say “new”, since I am talking about learning chemistry over 50 years ago!