Human tears largely consist of water, sodium, and potassium electrolytes. However, these are not the only important substances found in tears. There are a number of complex organic substances essential in maintaining the eye.
One ingredient is lysozyme, prominent among a group of chemicals which exhibit antibacterial behavior [see the PubChem reference for an image of lysozyme].
Lysozyme Modus Operandum
The University of Colorado reference (cited) makes the following statement: “The enzyme lysozyme breaks down bacterial cell walls, which are made of a unique compound called peptidoglycan.“
Molecular Structure of the Contenders
The fifty-cent name for lysozyme is N-acetylmuramide glycanhydrolase. The suffix at the end of the name, -ase, indicates this compound is an enzyme. An enzyme breaks down or digests something, as scissors cuts paper.
Peptidoglycan is alternately called murein. It is a polymer constructed from component sugars and amino acids. Check the images and see if you can spot these.
Specific Chemical Action
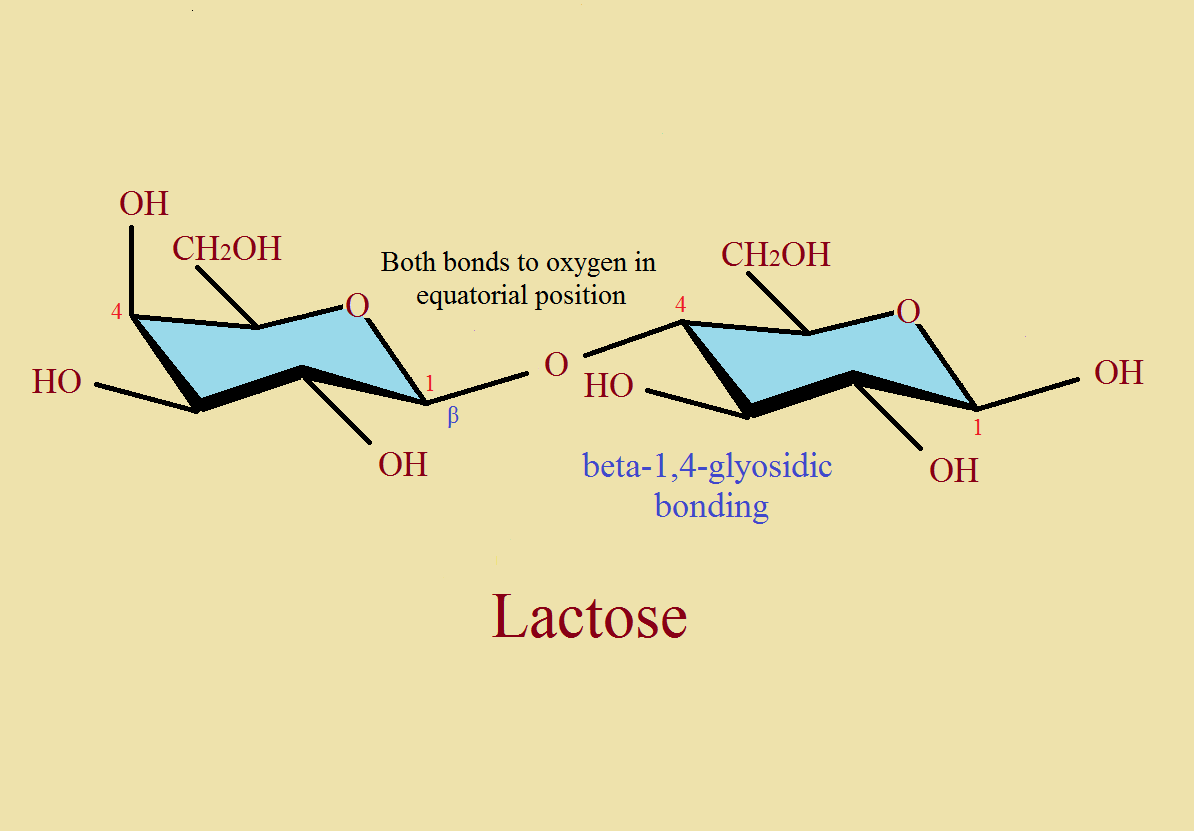
“The main action of lysozyme is to perform beta 1-4 glycosidic bond cleavage on the N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM) polysaccharide coating of a bacteria cell wall.” [reference cited] Let’s get acquainted with the participants. We begin with a little background information.
Background Information
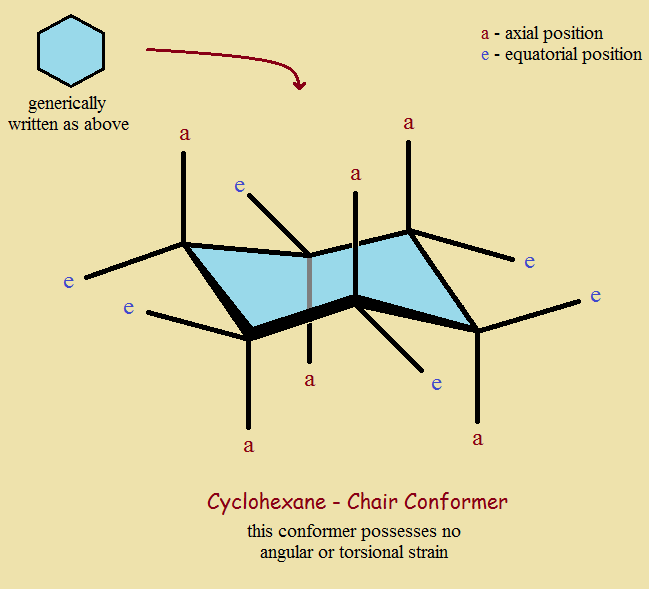
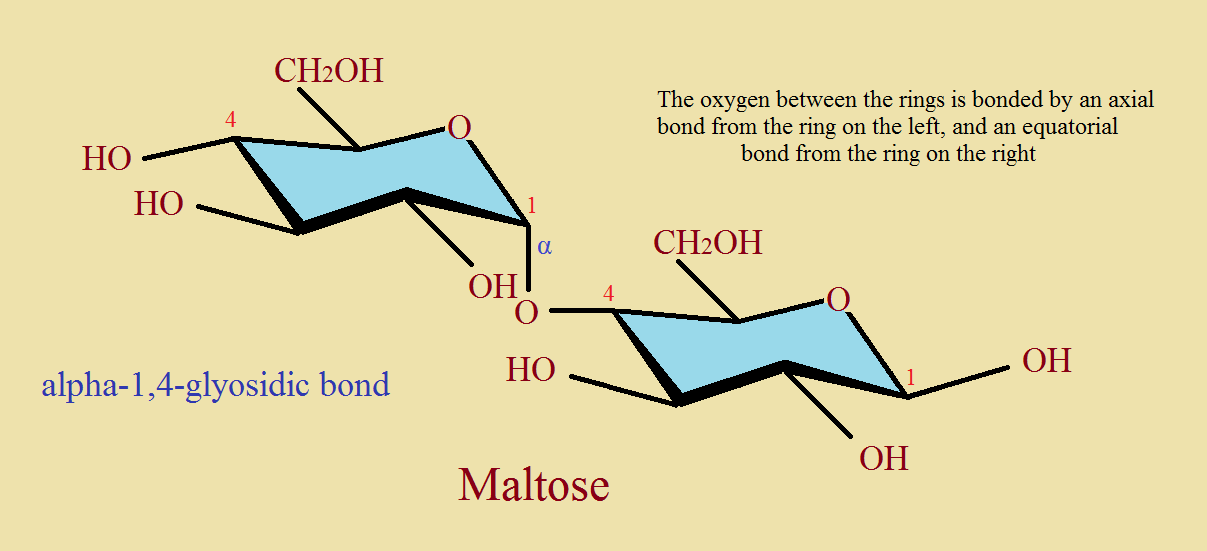
Notice among our images that a molecule of cyclohexane (C₆H₁₂) in its most stable “chair” conformer distributes its 12 attached hydrogen atoms in one of two positions, equatorial (around the circumference of the ring) and axial (above or below the plane of the ring).
Of course another atoms or group of atoms can replace hydrogen. Often if it is only hydrogen, equatorial or axial, that atom is not drawn to avoid “clutter”. Some ring structures do not consist only of carbon atoms. Sugars, for instance, may include an oxygen atom in the ring.
Second, notice what is meant by a β-1,4-glycosidic bond. Compare the two images, one of an α-bond, one with a β-bond between two 6-membered rings.
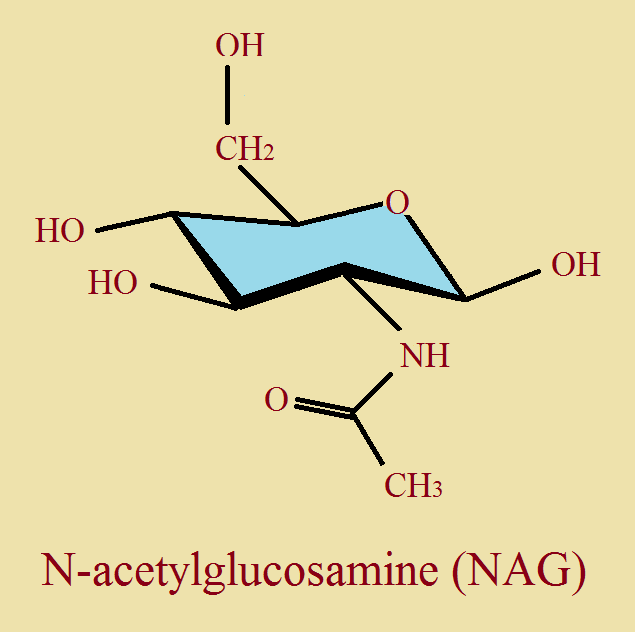
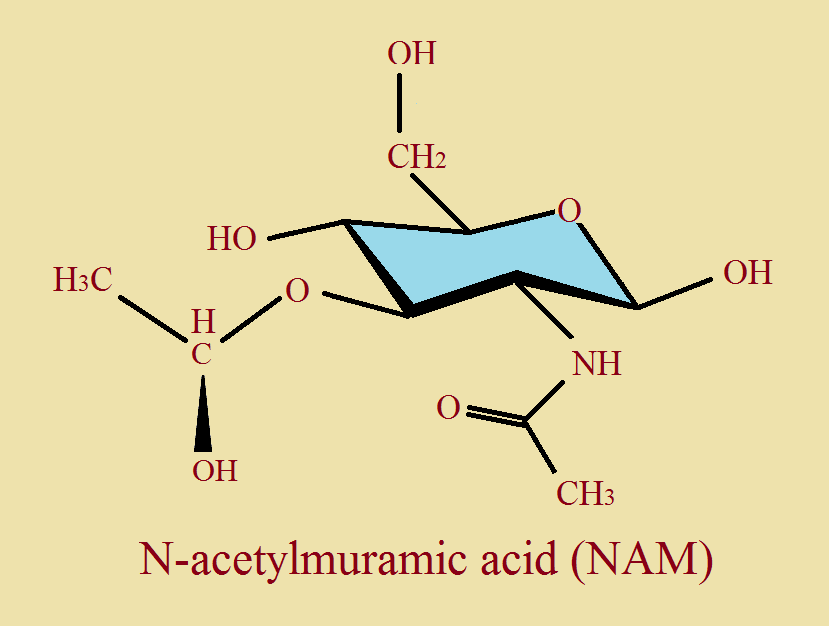
Third, note the image of NAG. As we have discussed, none of the axial hydrogen atoms have been drawn to avoid clutter. And the ring contains an oxygen in place of a carbon atom. The “n” in NAG stands for nitrogen. The ring, plus the nitrogen atom attached to it suggests glucose and amine. CH₃CO- is the acetyl group. The only difference between NAG and NAM is one modified hydroxyl group.
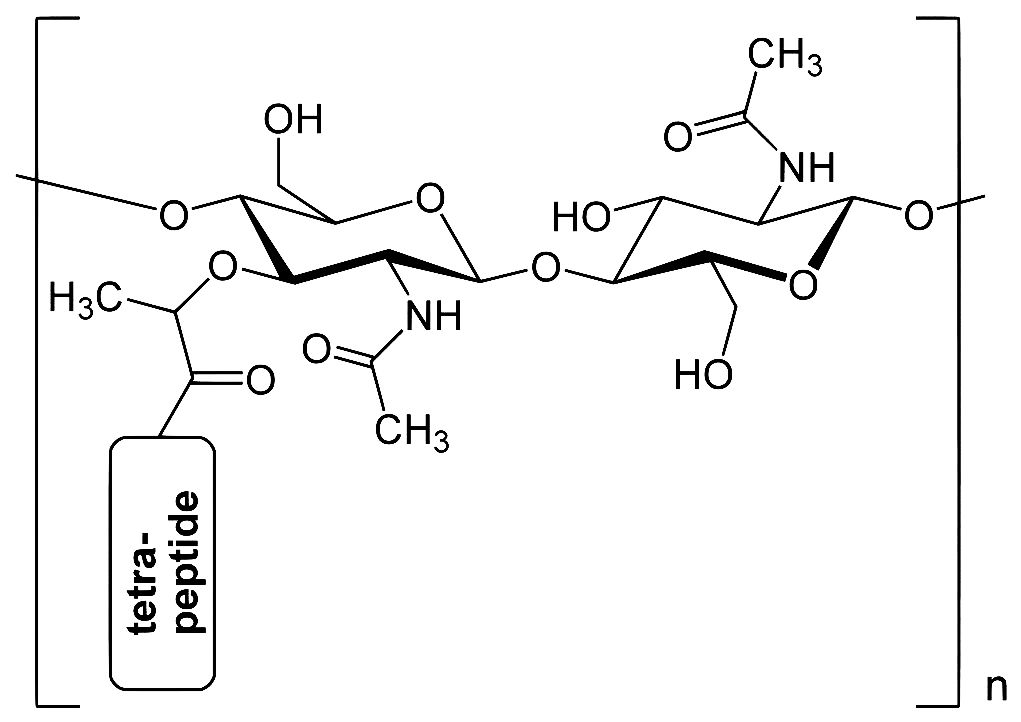
These units join together alternately to form a polysaccharide polymer. Then each of the NAM groups also join to peptide units, each 4 or 5 amino acids long [oligopeptide chains]. This complex structure form part of bacterial cell wall. And, to reiterate, this is our “Jericho” wall that must come ‘tumbling down’ under the influence of the tear ingredient, lysozyme.
It’s Pretty Amazing
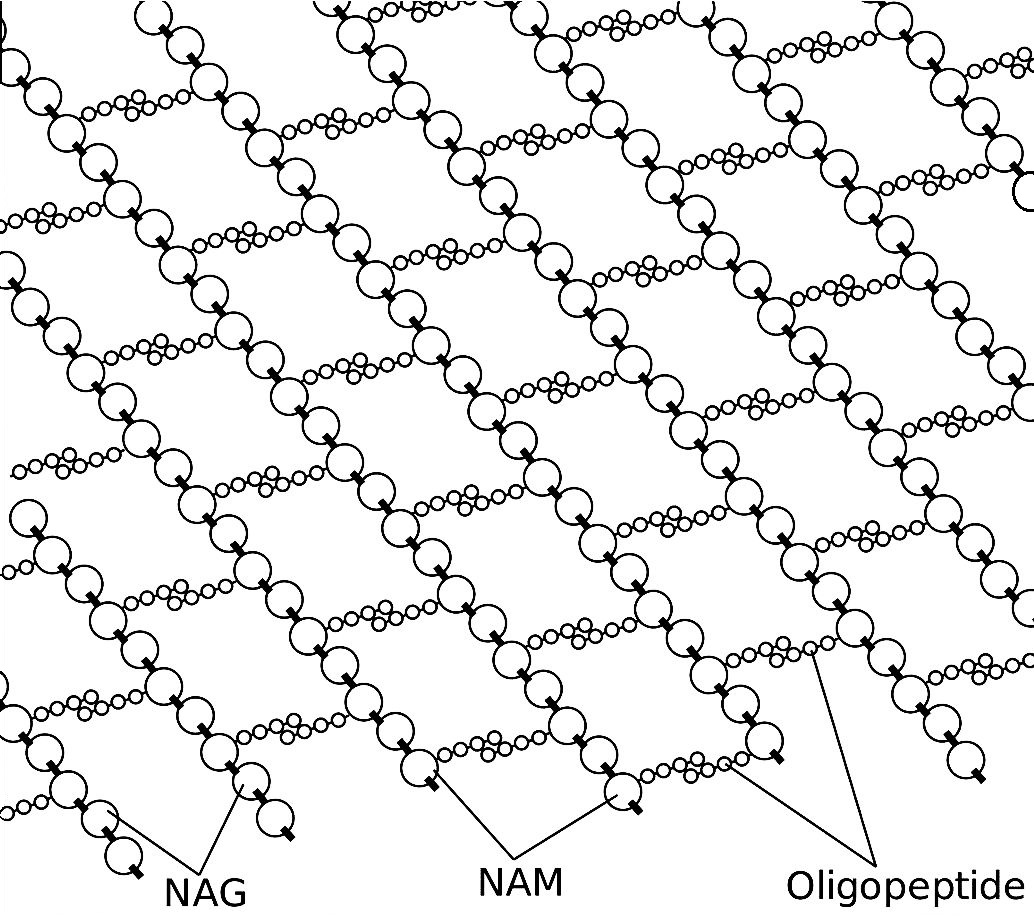
Precisely how the lysozyme snips the beta 1-4 glycosidic bonds is beyond the scope of this article. In fact, it is still undergoing investigation. But then, lysozyme in itself is pretty amazing. The two amazing videos that follow are about the topic of crystallography. However, they feature the example of (you guessed it) lysozyme.
Tear Ingredient Lysozyme Videos
Is such elegant chemistry as this (which is still not understood in its entirety) the product of an impersonal force? This is a science article – you decide.
Only, next time you rub your eyes, you will likely be more appreciative how those tears in your eyes are ‘in your corner’, fighting in your behalf.
Note: You might also enjoy How Far Can Perfect Eyes See? An Ideal Earthly Scenario
References:
- U. of Colorado: Biological Sciences Initiative: Lysozyme
- PubChem: Lysozyme (image)
- CA State Sci-Fair: Does Lysozyme in Human Tears Kill Bacteria?
- University of Wisconsin – Eau Claire: Human Lysozome
← Back to Food and Health
← Home

If you fail to watch the two short videos above, you will miss out on some of the fascination you should feel concerning even the simplest of life processes. I highly recommend watching them, even if you don’t understand much of what you will see…