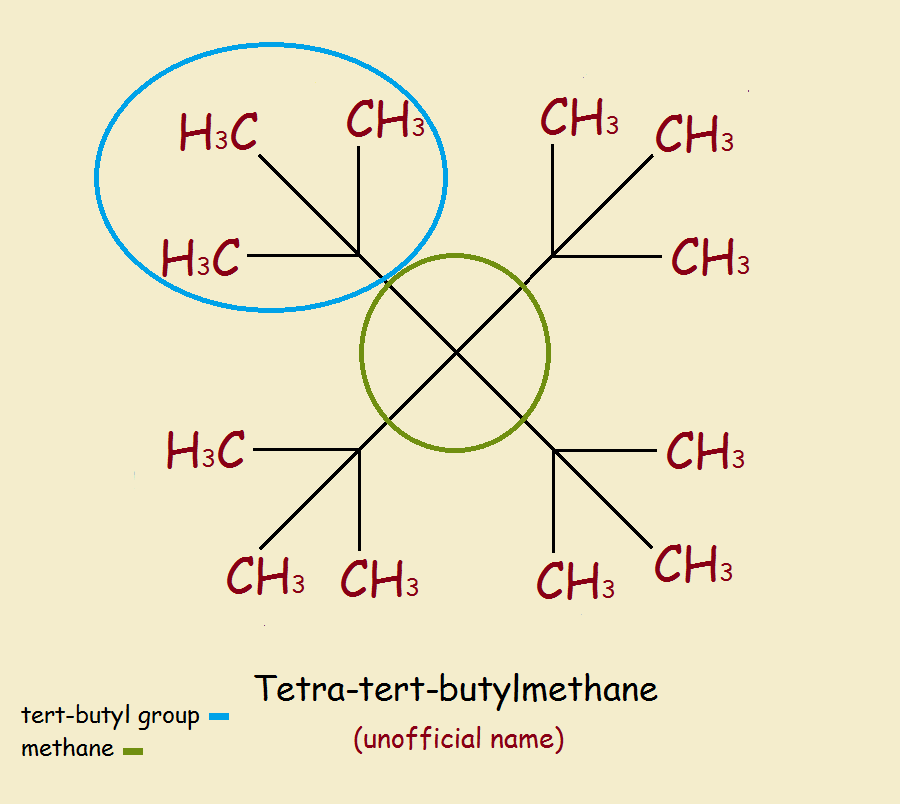
 The smallest non-cyclic alkane that theoretically should exist, but to date has never successfully been prepared, is tetra-tert-butyl methane. The reason it is so elusive is crowding or steric hindrance.
The smallest non-cyclic alkane that theoretically should exist, but to date has never successfully been prepared, is tetra-tert-butyl methane. The reason it is so elusive is crowding or steric hindrance.
Introducing TTBM
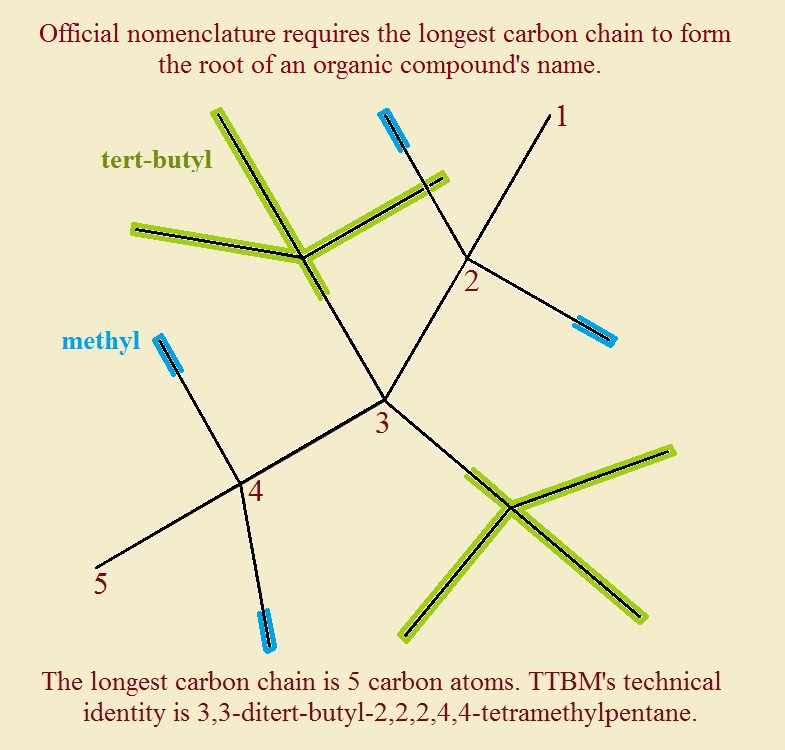
Tetra-tert-butyl methane is the common name of 3,3-ditert-butyl-2,2,4,4-tetramethylpentane.
A glimpse at the structure reveals the overcrowding. Despite the overcrowding, according to energy calculations, the molecule (we’ll call it TTBM) should be capable of existence.
Assembling the Model
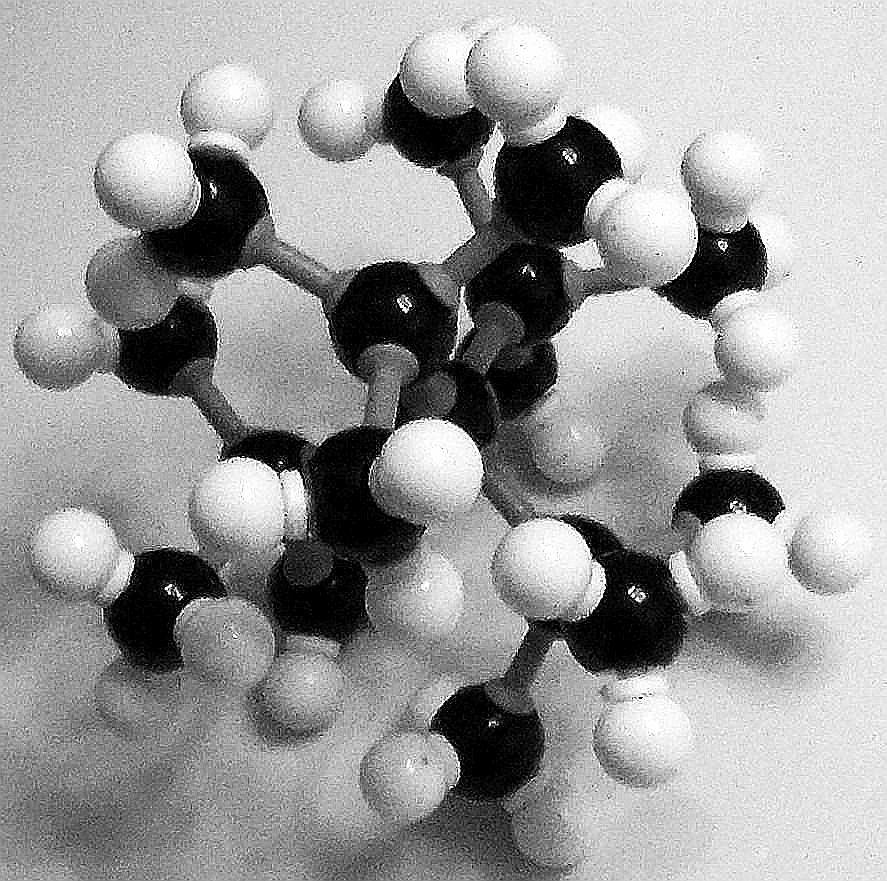
Even building the model from its component parts presented a measure of difficulty. The 17 carbon atoms (black spheres) and 36 hydrogen atoms (smaller white spheres) required some effort to assemble into TTBM. Still, it was accomplished with all the component atoms not touching one another. What is the problem or problems?
Synthesis
While attempts are still being made to prepare tetra-tert-butyl methane, it should be obvious that researchers have their work cut out for them. TTBM cannot be made by simple hand-assembly. Reagents are required. Reagent species of necessity are larger than the parts to be assembled.
Reactant molecules and molecular fragments need appropriate spatial orientation. Repulsive forces between already assembled atoms of the developing TTBM structure and additional reactant atoms make the task a most formidable one. It could be likened to assembling an interlocking puzzle while wearing boxing gloves.
Will Tetra-Tert-Butyl Methane Ever Be Synthesized?
 It is questionable if TTBM can be constructed. Even if it could be forced together, strain may force it to quickly disassemble. But maybe not. Certainly it will not fail to be synthesized because of lack of trying. If it can be made, it will be made is the mindset of Today’s scientist.
It is questionable if TTBM can be constructed. Even if it could be forced together, strain may force it to quickly disassemble. But maybe not. Certainly it will not fail to be synthesized because of lack of trying. If it can be made, it will be made is the mindset of Today’s scientist.
Note: You might also enjoy Hückel’s Smallest: The Aromatic Cyclopropenyl Cation
References:
- Journal of Chemical Information and Modeling: What Is the Smallest Saturated Acyclic Alkane that Cannot Be Made?
- The Journal of Physical Chemistry: Structural and Energetics Studies of Tri- and Tetra-tert-butylmethane
- March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure 6th Ed. p232

Makes me think of snowflakes for some reason, even though there are 12, ends, not 6. Every snowflake is said to be unique, maybe this molecule exists in some unique way as an interaction between other molecules of the same substance?