 When you go to the doctor for a flu shot, you may be asked to sign a waiver or release form. It indicates you no known negative responses to the presence of thimerosal. Perhaps you’ve never heard of this before? Well, perhaps you have under a different name. Many years ago it was called Merthiolate.
When you go to the doctor for a flu shot, you may be asked to sign a waiver or release form. It indicates you no known negative responses to the presence of thimerosal. Perhaps you’ve never heard of this before? Well, perhaps you have under a different name. Many years ago it was called Merthiolate.
Properties
Thimerosal is an antiseptic, antifungal compound. It is used as a preservative. It is also known as thiomersal. The prefix “thio” refers to the presence of sulfur. Thimerosal also contains an atom of mercury.
Structure
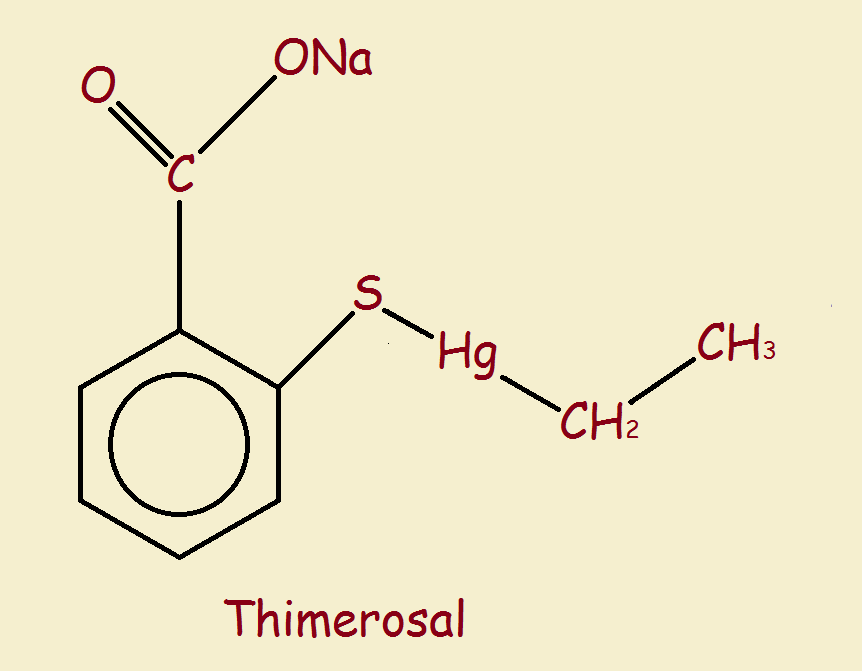
Its structure is seen in the image associated with this article. The S atom is sulfur, the Hg is hydrargyrum (mercury). The ring is a benzene ring, and the line segment to the right of the mercury atom is an ethyl group. The piece directly above the ring that extends to the right is the anionic carboxylate group with its attached sodium cation.
Metabolisis of Thimerosal
Note that the mercury atom is not part of the ionic (charged) portion of the molecule. Rather, metabolisis occurs between the mercury and sulfur atoms. This is seen from the resultant products, ethylmercury and thiosalicylate. The ethylmercury molecule is excreted by the gut in under a week.1
Controversy
Of course, it is the atom of mercury that is the primary source of controversy. However, mercury’s lack of ionicity makes excreting the mercury easier. It poses less of a hazard. Still, the use of thimerosal in vaccines has been greatly decreased within the U.S., especially when used to treat the very young.
Confusion and Conclusion
At one time, it was believed the preservative might be associated with autism. This has not been conclusively verified. Part of the concern arises because of confusing ethylmercury with methylmercury. The latter remains within the body for approximately a month and a half. It may then be absorbed by body membranes, where it converts to the ionic form of mercury.
So, although some minor, negative responses are experienced from its use, the mercury containing preservative may remain in use for a while longer.
1 World Health Organization: Global Advisory Committee on Vaccine Safety: Statement on Thiomersal.
Note: You might also enjoy The Deadly Morning Sickness Medication Thalidomide – Still in Use Today?
References:
