 In discussing the chemistry of the human body, the media features many important substances. Take water. We are told how important it is to drink lots of water. Our body cells do consist, after all, of about 60% water. And we repeatedly hear of DNA, cholesterol, free radicals, omega fatty-acids, trans fat and sodium.
In discussing the chemistry of the human body, the media features many important substances. Take water. We are told how important it is to drink lots of water. Our body cells do consist, after all, of about 60% water. And we repeatedly hear of DNA, cholesterol, free radicals, omega fatty-acids, trans fat and sodium.
Yet, there is one essential compound that is seldom mentioned… adenosine triphosphate, acronym ATP. Have you heard-tell of ATP? No? Yet it is remarkably important – you could not exist without it. Besides, even if you could, the energy you’d need to function would be totally unavailable.
ATP – What Is It?
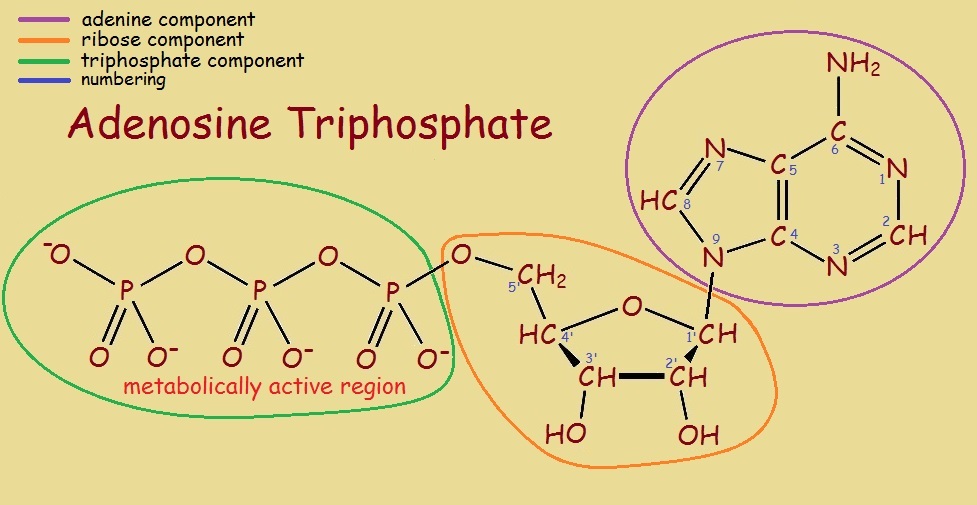
ATP, on examination (see image) is one molecule consisting of three portions. There is an adenine portion attached to a ribose (sugar) portion, attached to a triphosphate portion. Perhaps the reader recognizes these are components also found in deoxyribosenucleic acid, or DNA (see DNA: The Modular Staircase of Life).
For our purposes, ATP is like a battery, storing energy, that is easily tapped, then restored, chemically, much as a battery is restored by an alternator. The first part of the process is described, below.
ATP – Where Is It Found?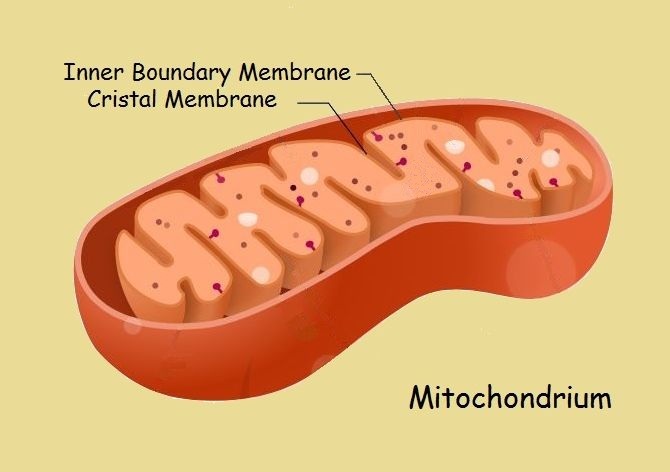
In what cells, and in what parts of the cells may adenosine triphosphate be found? It is found (and synthesized) in the membrane of mitchondria. OK. What are mitochondria? They are small organelles within the cell that are particularly associated with, among other things, ATP production, cell differentiaton, and cell death.
Source of Adenosine Triphosphate’s Energy
How is it that adenosine triphosphate can provide energy to fuel metabolic processes? The phosphoryl groups (constituting the metabolically active region) each possess a high-energy bond that is readily broken, yielding adenosine diphosphate (ADP). Occasionally, two phosphoryl groups broken off, yielding adenosine monophosphate. Energy is released.
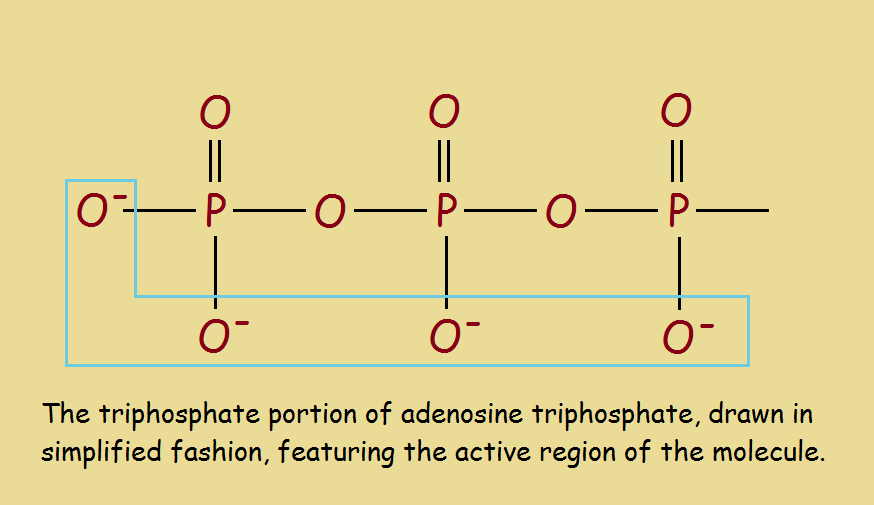
So we can write:
ATP → ADP + Energy
and
ATP → AMP + Energy
Extra Credit:
Why not enjoy this brief video (6:18) providing additional details on what we have discussed?¹
1 Note that the phosphorous and oxygen containing groups attached to ATP are referred to as phosphoryl groups. When they leave, they react to form ionic phosphate groups.
Note: You might also enjoy Mono- Di- and Polysaccharides: Starches Sugars and Cellulose
References:
- USGS: The Water in You: Water and the Human Body
- Georgia State University – Hyperphysics: Adenosine Triphosphate
← Back to Classic Science
← Home

Long ago biology lessons ATP and ADP! We never had such nice diagrams, though.
Thank you so much!